5557. Lofepramine
Nomenclature
CAS number: 23047-25-8
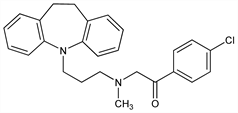
1-(4-Chlorophenyl)-2-[[3-(10,11-dihydro-5H-dibenz[b,f]azepin-5-yl)propyl]methylamino]ethanone; 4′-chloro-2-[[3-(10,11-dihydro-5H-dibenz[b,f]azepin-5-yl)propyl]methylamino]acetophenone; N-methyl-N-(4-chlorobenzoylmethyl)-3-(10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)propylamine; lopramine. C26H27ClN2O; mol wt 418.96.
C 74.54%, H 6.50%, Cl 8.46%, N 6.69%, O 3.82%.
Description and references
Psychotropic drug related to imipramine, q.v. Prepn: E. Eriksoo et al., GB 1177525; eidem, US 3637660 (1970, 1972 both to AB Leo). Chemistry and pharmacology: E. Eriksoo, O. Rohte, Arzneim.-Forsch. 20, 1561 (1970). Absorption and metabolism: J. R. Tulic et al., Acta Pharmacol. Toxicol. 32, 304 (1973). Distribution and excretion: G. Plym Forshell, Xenobiotica 5, 73 (1975). Pharmacokinetics: G. Plym Forshell et al., Eur. J. Clin. Pharmacol. 9, 291 (1976). Clinical study: S. Wright, L. Herrmann, Arzneim.-Forsch. 26, 1167 (1976). Review of clinical experience in depression: S. G. Lancaster, J. P. Gonzalez, Drugs 37, 123-140 (1989).
Properties
Crystals from methanol or acetone, mp 104-106°. Easily oxidized by air and other oxidizing agents to desipramine and p-chlorobenzoic acid.Derivative
Hydrochloride.Nomenclature
CAS number: 26786-32-3
Leo 640; Amplit (Daiichi); Gamanil (Lundbeck); Gamonil (Merck KGaA); Lomont (Rosemont); Tymelyt (Lundbeck). C26H27ClN2O.HCl; mol wt 455.42.
C 68.57%, H 6.20%, Cl 15.57%, N 6.15%, O 3.51%.
Properties
Crystals from butanone, mp 152-154°. Sol in methanol, ethanol, chloroform. Practically insol in water. LD50 in mice, rats (mg/kg): >2500, >1000 orally; 920, >1000 i.p.; >1000, >1000 s.c. (Eriksoo, Rohte).Therapeutic Category
Antidepressant.
Keywords
Antidepressant; Tricyclics