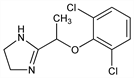
5558. Lofexidine
Nomenclature
Description and references
α2-Adrenoceptor agonist related structurally to clonidine, q.v. Prepn of the HCl salt: H. Baganz, H. J. May, ZA 6800850; eidem, US 3966757 (1968, 1976 both to Nordmark); of the free base: eidem, DE 1935479 (1971 to Nordmark), C.A. 74, 87979 (1971). Pharmacological studies: J. Velly, J. Pharmacol. 8, 351 (1977); B. Jarrot et al., Biochem. Pharmacol. 28, 141 (1979). NMR data and cardiovascular effects: P. B. M. Timmermans, P. A. Van Zwieten, Eur. J. Med. Chem. 15, 323 (1980). Hypotensive and sedative properties: P. Birch et al., Br. J. Pharmacol. 68, 107 (1980). Effects in hypertension: N. D. Vlachakis et al., Fed. Proc. 39, 4844 (1980). Series of articles on pharmacology, toxicology, clinical studies: Arzneim.-Forsch. 32, 915-993 (1982). Toxicity studies: T. H. Tsai et al., ibid. 955. Review of clinical trials in treatment of opiate withdrawal: J. Strang et al., Am. J. Addict. 8, 337-348 (1999).
Properties
Crystals, mp 126-128°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol/ether or 2-propanol, mp 221-223° (U.S. patent); also reported as mp 230-232° (Ger. patent). Very sol in water, ethanol. Slightly sol in 2-propanol. Practically insol in ether. LD50 in mice, rats, dogs (mg/kg): between 74-147 orally (all species); between 8-18 i.v. (all species) (Tsai).Therapeutic Category
In treatment of opioid withdrawal symptoms; antihypertensive.
Keywords
Antihypertensive; Imidazole Derivatives