5778. Meclocycline
Nomenclature
CAS number: 2013-58-3
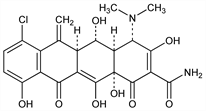
[4S-(4α,4aα,5α,5aα,12aα)]-7-Chloro-4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methylene-1,11-dioxo-2-naphthacenecarboxamide; 7-chloro-6-methylene-5-hydroxytetracycline; GS-2989; NSC-78502. C22H21ClN2O8; mol wt 476.86.
C 55.41%, H 4.44%, Cl 7.43%, N 5.87%, O 26.84%.
Description and references
Semi-synthetic antibiotic derived from tetracycline, q.v. Prepn: R. K. Blackwood et al., US 2984686 (1961 to Pfizer); eidem, J. Am. Chem. Soc. 83, 2773 (1961); of the free base and 5-sulfosalicylate salt: eidem, ibid. 85, 3943 (1963). In vitro study: L. Lucca, G. Vittadini, G. Ital. Chemioter. 26, 203 (1979). 13C-NMR study: E. Mazzola et al., J. Pharm. Sci. 69, 229 (1980). Toxicity study: F. Bernardi et al., G. Ital. Chemioter. 17, 276 (1970).
Properties
uv max (methanol, 0.01N HCl): 245, 347 nm (log ε 4.34, 410). LD50 in mice (mg/kg): >5000 orally, 425 i.p. (Bernardi).Derivative
5-Sulfosalicylate.Nomenclature
CAS number: 73816-42-9
Meclan (Ortho); Mecloderm; Meclosorb (Basotherm); Meclutin (ABC); Traumatociclina (Biomed. Foscama). C29H27ClN2O14S; mol wt 695.05.
C 50.11%, H 3.92%, Cl 5.10%, N 4.03%, O 32.23%, S 4.61%.
Properties
uv max (methanol, 0.01N HCl): 239, 268, 346 nm (log ε 4.46, 4.07, 4.11).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Tetracyclines