5831. Menadione
Nomenclature
Description and references
Synthetic naphthoquinone derivative having physiologic properties of vitamin K, q.v. Prepn: Fieser, J. Biol. Chem. 133, 391 (1940). Toxicity study: Molitor, Robinson, Proc. Soc. Exp. Biol. Med. 43, 725 (1940). Alkylated in vivo to the bioactive metabolite, menaquinone-4, q.v.: C. Martius, H. O. Esser, Biochem. Z. 331, 1 (1958). Metabolism and tissue distribution: W. V. Taggart, J. T. Matschiner, Biochemistry 8, 1141 (1969). Improved synthesis: W. Adam et al., Angew. Chem. Int. Ed. 33, 2475 (1994).
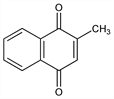
Properties
Bright yellow crystals. Very faint acrid odor. mp 105-107°. Stable in air; dec by sunlight. Insol in water. One gram dissolves in about 60 ml alcohol, in 10 ml benzene, in 50 ml vegetable oils; moderately sol in chloroform, carbon tetrachloride. The alcoholic soln is neutral to litmus. Solutions may be heated to 120° without dec. Destroyed by alkalies and reducing agents. Keep protected from light. LD50 orally in mice: ≈0.5 g/kg (Molitor, Robinson).Derivative
Sodium bisulfite.Nomenclature
Description and references
Prepn: Moore, J. Am. Chem. Soc. 63, 2049 (1941). Structure: Carmack et al., ibid. 72, 844 (1950).
Properties
Occurs as the trihydrate. White, hygroscopic crystals. Discolors and may turn purple under the influence of light. One gram dissolves in ≈2 ml water. Slightly sol in alcohol. Almost insol in ether, benzene.Derivative
Dimethylpyrimidinol bisulfite.Nomenclature
Description and references
Prepn: J. B. Nanninga et al., US 3328169 (1967 to Heterochemical). Efficacy in swine diets: R. W. Seerley et al., J. Anim. Sci. 42, 599 (1976).
Properties
Cryst powder, mp 215-217°. Soly in water ≈1 g/100 ml. Slightly sol in alcohol. Insol in ether, benzene.Therapeutic Category
Vitamin (prothrombogenic).
Therapeutic Category (Veterinary)
Vitamin (prothrombogenic); antidote to bishydroxycoumarin poisoning, including sweet clover poisoning.Keywords
Vitamin/Vitamin Source; Vitamin K