5833. Menaquinones
Nomenclature
Vitamin K2; 2-methyl-3-all-trans-polyprenyl-1,4-naphthoquinones.Description and references
Group of prenylated naphthoquinone derivatives having the physiological activity of vitamin K, q.v. Nomenclature is based on the number of isoprene residues comprising the side chain. Compds of varying chain length are produced by bacteria, particularly by normal intestinal flora, and may serve as a source of vitamin K for humans. Birds and animals are capable of alkylating menadione, q.v., to produce menaquinone 4. Originally isolated from putrefied fish meal: R. W. McKee et al., J. Biol. Chem. 131, 327 (1939). Identification of menaquinone 6: S. B. Binkley et al., ibid. 133, 721 (1940). Isoln of menaquinone 7 from cultures of Bacillus brevis: M. Tishler, W. L. Sampson, Proc. Soc. Exp. Biol. Med. 68, 136 (1948). Structures and syntheses of series of menaquinones: O. Isler et al., Helv. Chim. Acta 41, 786 (1958). Review of isoln, props, synthesis: O. Isler, O. Wiss, Vitam. Horm. 17, 53-90 (1959); H. Mayer, O. Isler, Methods Enzymol. 18C, 469-547 (1971); of biosynthesis: D. R. Threlfall, Vitam. Horm. 29, 153-200 (1971); R. Bentley, R. Meganathan, Microbiol. Rev. 46, 241-280 (1982). Menaquinones with side chains of up to 15 repeating units have been described: T. Sakano et al., Chem. Pharm. Bull. 34, 4322 (1986). HPLC determn in bone: S. J. Hodges et al., J. Bone Miner. Res. 8, 1005 (1993). Metabolism and distribution: H. H. W. Thijssen, M. J. Drittij-Reijnders, Br. J. Nutr. 72, 415 (1994). Absorption and bioavailability in humans: J. M. Conly et al., Am. J. Gastroenterol. 89, 915 (1994).
Derivative
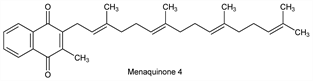
Menaquinone 4.Nomenclature
Properties
Yellow crystals from alcohol, mp 35°. uv max: 248 nm (E1%1cm 439).Derivative
Menaquinone 6.Nomenclature
Properties
Yellow crystals from acetone + alcohol or petr ether, mp 50°. uv max (petr ether): 243, 248, 261, 270, 325-328 nm (E1%1cm 304, 320, 290, 292, 53).Derivative
Menaquinone 7.Nomenclature
Properties
Light yellow microcrystalline plates from petr ether, mp 54°. bp0.0002 200° (some dec). uv max (petr ether): 243, 248, 261, 270, 325-328 nm (E1%1cm 278, 195, 266, 267, 48). Slightly less sol than vitamin K1 in the same organic solvents.Therapeutic Category
Vitamin (prothrombogenic).
Keywords
Vitamin/Vitamin Source; Vitamin K