5901. Mersalyl
Nomenclature
CAS number: 492-18-2
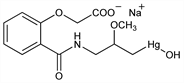
[3-[[2-(Carboxylatomethoxy)benzoyl]amino]-2-methoxypropyl]hydroxymercurate(1)
sodium; o-[[3-(hydroxymercuri)-2-methoxypropyl]carbamoyl]phenoxyacetic
acid sodium salt; sodium o-[(3-hydroxymercuri-2-methoxypropyl)carbamoyl]phenoxyacetate; N-(γ-hydroxymercuri-β-methoxypropyl)salicylamide-O-acetic acid sodium salt; mercuramide; Salyrgan (Sterling Winthrop). C13H16HgNNaO6; mol wt 505.85.
C 30.87%, H 3.19%, Hg 39.65%, N 2.77%, Na 4.54%, O 18.98%.
Description and references
Prepn: Diels, Beccard, Ber. 39, 4125 (1906); Bockmühl, Schwarz, DE 423031 (1925 to Hoechst); Frdl. 15, 1609. Use as diuretic: F. Brunn, Wien. Klin. Wochenschr. 37, 901 (1924). Toxicity data: E. B. Robbins, K. K. Chen, J. Am. Pharm. Assoc. 40, 249 (1951). Use of acid as physiological sulfhydryl inhibitor: P. Mavier, J. Hanoune, Eur. J. Biochem. 59, 593 (1975).
Properties
Bitter crystals. Somewhat deliquescent. Gradually dec by light. One gram dissolves in about 1 ml water, in about 3 ml ethanol (95%), in 2 ml abs methanol. Practically insol in ether, chloroform. Aq solns are alkaline to litmus. LD50 i.v. in mice, rats: 72.6, 17.7 mg/kg (Robbins, Chen).Derivative
Mersalyl Acid.Nomenclature
CAS number: 486-67-9
C13H17HgNO6; mol wt 483.87.
C 32.27%, H 3.54%, Hg 41.46%, N 2.89%, O 19.84%.
Properties
Odorless, white, slightly hygroscopic powder. Slightly sol in water, dil mineral acids; sol in solutions of alkali hydroxides.Use
Sulfhydryl specific biochemical probe.Therapeutic Category
Diuretic.
Therapeutic Category (Veterinary)
Diuretic.Keywords
Diuretic; Organomercurials