6091. Methylisothiazolinone
Nomenclature
CAS number: 2682-20-4
2-Methyl-3(2H)-isothiazolone; MI; MIT; Neolone 950 (Rohm & Haas). C4H5NOS; mol wt 115.15.
C 41.72%, H 4.38%, N 12.16%, O 13.89%, S 27.85%.
Description and references
Marketed in combination with methylchloroisothiazolinone as a broad spectrum biocide. Prepn: W. D. Crow, N. J. Leonard, Tetrahedron Lett. 5, 1477 (1964); eidem, J. Org. Chem. 30, 2660 (1965); S. N. Lewis et al., J. Heterocycl. Chem. 8, 571 (1971). HPLC determn in cosmetic products: R. Matissek, Chromatographia 28, 34 (1989). In vitro neurotoxicity study: S. Du et al., J. Neurosci. 22, 7408 (2002). Safety assessment: J. Am. Coll. Toxicol. 11, 75-128 (1992). Review of epidemiology of allergic reactions: A. C. de Groot, J. W. Weyland, J. Am. Acad. Dermatol. 18, 350-358 (1988) J. Fewings, T. Menné, Contact Dermatitis 41, 1-13 (1999).
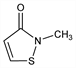
Properties
Colorless prisms, mp 50-51°. bp0.03 93°. uv max (diethyl ether): 281 nm (ε 6550). uv max (95% ethanol): 275 nm (ε 7250).Derivative
Methylchloroisothiazolinone.Nomenclature
CAS number: 26172-55-4
5-Chloro-2-methyl-3(2H)-isothiazolone; MCI. C4H4ClNOS; mol wt 149.60.
C 32.11%, H 2.70%, Cl 23.70%, N 9.36%, O 10.69%, S 21.43%.
Properties
Crystals from ligroin (60-90°), mp 54-55°. uv max (methanol): 277 nm (log ε 3.82).Derivative
Mixture with methylchloroisothiazolinone.Nomenclature
CAS number: 55965-84-9
Kathon CG (Rohm & Haas).