Nomenclature
CAS number: 872-50-4
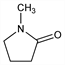
1-Methyl-2-pyrrolidinone;
N-methyl-α-pyrrolidinone;
N-methyl-γ-butyrolactone; NMP; 1-methylazacyclopentan-2-one; MP; M-Pyrol (ISP).
C
5H
9NO; mol wt 99.13.
C 60.58%, H 9.15%, N 14.13%, O 16.14%.
Description and references
Dipolar aprotic solvent. Commercially prepd
by condensation of butyrolactone with methylamine. Prepn: J. Tafel,
O. Wassmuth, Ber. 40, 2831 (1907); E. Spth, J. Lintner, ibid. 69, 2727 (1936). Use in extraction of unsaturated/aromatic
compounds: E. Mueller, G. Hoehfeld, Proc.
8th World Pet. Congr. 4 (Applied Sci. Publishers,
London, 1971) pp 213-219; H. Klein, H. M. Weitz, Hydrocarbon Process. 47, 135 (November, 1968);
A. Sequeira et al., ibid. 58, 155 (September, 1979). Physical properties and solubility:
Industrial Solvents Handbook (3rd Ed.) 580-582 (1985).
Reference guide to properties and use: M-Pyrol, N-Methyl-2-Pyrrolidone
Handbook, ISP (subsidiary of GAF Corp.), 1988. Acute toxicity:
W. Bartsch et al., Arzneim.-Forsch. 26, 1581 (1976). Review: E. V. Hort, L.
R. Anderson in Kirk-Othmer Encyclopedia of
Chemical Technology vol. 19 (Wiley-Interscience,
New York, 3rd ed., 1982) pp 514-520.
Properties
Clear liquid. mp 24.4°. bp760 202°. nD25 1.4690. d425 1.027. Flash point, closed cup: 199°F; open cup: 204°F. Viscosity (25°): 1.65 cP. Surface tension at 25°: 40.7
dynes/cm. Dipole moment at 25°: 4.09 D. Dielectric constant at
25°: 32.2. Vapor pressure at 23.2°, 65°: 0.33, 5 mm Hg. Heat of
combustion: 719 kcal/mol; of vaporization: 127.3 kcal/kg. Misc
with water, alcohol, ether, acetone, ethyl acetate, chloroform, benzene.
LD50 in mice, rats (ml/kg): 3.5, 2.2 i.v.; 4.3. 2.4 i.p.; 7.5, 3.8 orally (Bartsch).Use
Industrial solvent primarily used in the extraction
of aromatics from lube oils. Other applications include recovery
and purification of acetylenes, olefins and diolefins, gas purification,
and aromatics extraction from feedstocks. Also, used as polymer solvent.
