6137. Methysergide
Nomenclature
CAS number: 361-37-5
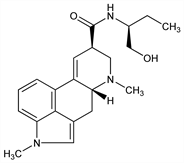
(8β)-9,10-Didehydro-N-[(1S)-1-(hydroxymethyl)propyl]-1,6-dimethylergoline-8-carboxamide; N-[1-(hydroxymethyl)propyl]-1-methyl-d-lysergamide; 1-methylmethylergonovine; 1-methyl-d-lysergic acid butanolamide; 1-methyl-d-lysergic acid (+)-1-hydroxy-2-butylamide; UML-491. C21H27N3O2; mol wt 353.46.
C 71.36%, H 7.70%, N 11.89%, O 9.05%.
Description and references
Serotonin receptor antagonist. Prepn: GB 854569 (1960); A. Hofmann, F. Troxler, US 3113133 (1963 to Sandoz). Comparative pharmacology: Z. Votava et al., Arzneim.-Forsch. 16, 220 (1966); P. N. Chambers, P. B. Marshall, J. Pharm. Pharmacol. 19, 65 (1967). Mechanism of action: D. A. Curran et al., Res. Clin. Stud. Headache 1, 74 (1967); J. E. Hardebo et al., Neurology 28, 64 (1978); S. W. J. Lamberts, R. M. Mac Leod, Endocrinology 103, 287 (1978).
Properties
Crystals, mp 194-196°. [α]D20 45° (c = 0.5 in pyridine).Derivative
Tartrate.C46H60N6O10; mol wt 857.00.
C 64.47%, H 7.06%, N 9.81%, O 18.67%.
Properties
Crystals, sparingly sol in water.Derivative
Dimaleate.Properties
Dec ≈165°. Sol in methanol, less sol in water (1:250). Practically insol in abs ethanol.Derivative
Hydrogen maleate.Nomenclature
CAS number: 129-49-7
Methysergide maleate; Deseril (Novartis); Désernil (Novartis); Sansert (Novartis). C21H27N3O2.C4H4O4; mol wt 469.53.
C 63.95%, H 6.65%, N 8.95%, O 20.45%.
Therapeutic Category
Antimigraine.
Keywords
Antimigraine; Serotonin Receptor Agonist; Serotonin Receptor Antagonist