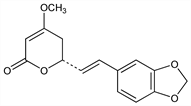
6138. Methysticin
Nomenclature
Description and references
One of the active components of the intoxicating beverage Kava from the Pacific Islands. From root of Piper methysticum Forst., Piperaceae (kava): Pomeranz, Monatsh. Chem. 10, 783 (1889); Hnsel, Beiersdorff, Arzneim.-Forsch. 9, 581 (1955). Structure: Borsche et al., Ber. 60, 2113 (1927). Synthesis of racemate: Klohs et al., J. Org. Chem. 24, 1829 (1959). Absolute config: Snatzke, Hnsel, Tetrahedron Lett. 1968, 1797. Crystal structure: P. Engel, W. Nowacki, Z. Kristallogr. 136, 437 (1972). 13C NMR: A. Banerji et al., Org. Magn. Reson. 13, 345 (1980). HPLC determn: R. M. Smith et al., J. Chromatogr. 283, 303 (1984). Anticonvulsant effects: Kretzschmar, Meyer, Arch. Int. Pharmacodyn. Ther. 177, 261 (1969). Neuroprotective activity: C. Backhauss, J. Krieglstein, Eur. J. Pharmacol. 215, 265 (1992).