6215. Mitomycins
Nomenclature
Description and references
A group of antitumor antibiotics produced by Streptomyces caespitosus (griseovinaceseus). Isoln of mitomycins A and B: T. Hata et al., J. Antibiot. 9A, 141 (1956). Isoln of C: S. Wakaki et al., Antibiot. Chemother. 8, 228 (1958); GB 830874; T. Hata et al., US 3660578 (1960, 1972 both to Kyowa and Kitasato Kenkyusho). Production of the complex: T. Yamamoto, H. Umezawa, JP 56 2898 (1956 to Nippon Antibiot. Subs. Sci. Assoc.), C.A. 51, 9100 (1957). Improved method: A. Gourevitch et al., US 3042582 (1962 to Bristol-Myers). Isoln from S. verticillatus, constituents: D. V. Lefemine et al., J. Am. Chem. Soc. 84, 3184 (1962). Structures: J. S. Webb et al., ibid. 3185, 3187. Total synthesis of mitomycins A and C: T. Fukuyama et al., Tetrahedron Lett. 1977, 4295. Toxicity: S. Kinoshita et al., J. Med. Chem. 14, 13 (1971). Review and evaluation of studies of carcinogenic action of mitomycin C in laboratory animals: IARC Monographs 10, 171-179 (1976). Review of syntheses: Y. Kishi, J. Nat. Prod. 42, 549-568 (1979). General review: R. W. Franck, Fortschr. Chem. Org. Naturst. 38, 1-41 (1979).
Derivative
Mitomycin A.Nomenclature
Description and references
The 6-methoxy analog of mitomycin C. X-ray crystallographic study: A. Tulinsky, J. Am. Chem. Soc. 84, 3188 (1962); A. Tulinsky, J. H. van den Hende, ibid. 89, 2905 (1967).
Properties
Red-violet crystals from acetone + carbon tetrachloride, dec 159-161°. Absorption max (water): 215, 318, 530 nm (E1%1cm 234, 122, 118.8). Sol in water, benzene, toluene, trichloroethylene, nitrobenzene and many organic solvents. Practically insol in xylene, carbon tetrachloride, carbon disulfide, petr ether, ligroin, cyclohexane. LD50 i.v. in mice: 2 mg/kg (Hata, 1956; Kinoshita).Derivative
Mitomycin B.Nomenclature
Description and references
Molecular structure: R. Yahashi, I. Matsubara, J. Antibiot. 29, 104 (1976); ibid. 31(6), correction (1978).
Properties
Violet crystals from acetone + carbon tetrachloride, dec 182-184°. Absorption max (water): 220, 320, 550 nm (E1%1cm 117.5, 55.0, 9.9). Sol in water and many organic solvents. Practically insol in xylene, carbon tetrachloride, carbon disulfide, petr ether, ligroin, cyclohexane, benzene, toluene, trichloroethylene, nitrobenzene. LD50 i.v. in mice: 10 mg/kg (Hata, 1956); also reported as 3 mg/kg (Kinoshita).Derivative
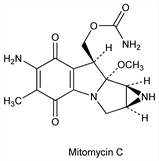
Mitomycin C.Nomenclature
Description and references
Chemistry and structure: C. L. Stevens et al., J. Med. Chem. 8, 1 (1965). Crystal and molecular structure: K. Ogawa et al., Bull. Chem. Soc. Jpn. 52, 2334 (1979). Revised absolute configuration: K. Shirahata, N. Hirayama, J. Am. Chem. Soc. 105, 7199 (1983). Mechanism of action: C. Rodigheriero, Farmaco Ed. Sci. 33, 651 (1978). Comprehensive description: J. H. Beijnen et al., Anal. Profiles Drug Subs. 16, 361-401 (1986).
Properties
Blue-violet crystals, does not melt below 360°. Absorption max (methanol): 216, 360, 560 nm (E1%1cm 742, 742, 0.06). Sol in water, methanol, acetone, butyl acetate and cyclohexanone; slightly sol in benzene, carbon tetrachloride, ether. Practically insol in petr ether. LD50 i.v. in mice: 5 mg/kg (Wakaki); also reported as 9 mg/kg (Kinoshita).Derivative
N-Methylmitomycin C see Porfiromycin.Therapeutic Category
Mitomycin C as antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs