7597. Porfiromycin
Nomenclature
CAS number: 801-52-5
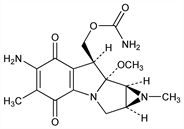
[1aS-(1aα,8β,8aα,8bα)]-6-Amino-8-[[(aminocarbonyl)oxy]methyl]-1,1a,2,8,8a,8b-hexahydro-8a-methoxy-1,5-dimethylazirino[2′,3′:3,4]pyrrolo[1,2-a]indole-4,7-dione; N-methylmitomycin C; U-14743. C16H20N4O5; mol wt 348.35.
C 55.17%, H 5.79%, N 16.08%, O 22.96%.
Description and references
Antibiotic substance isolated from a Streptomyces ardus fermentation broth: Herr et al., Antimicrob. Agents Annu. 1960, 23. Isoln from S. verticillatus and structure: Webb et al., J. Am. Chem. Soc. 84, 3185, 3187 (1962). Production process: Bohonos et al., US 3219530 (1965 to Am. Cyanamid). Synthesis of (±)-form: F. Nakatsubo et al., J. Am. Chem. Soc. 99, 8115 (1977). For stereochemistry and other synthetic studies, see Mitomycins.
Properties
Dark purple triclinic crystals, dec 201-201.5°. [α]D25 +275 ±55° (c = 0.1% in methanol); [α]D25 +242 ±100° (c = 0.045% in methanol). uv max (methanol): 217, 360, 555 nm (ε 24600, 23000, 209). Slightly sol in water, moderately sol in polar organic solvents. Practically insol in hydrocarbon solvents.Therapeutic Category
Antibacterial; antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs