6328. Mycosamine
Nomenclature
CAS number: 527-38-8
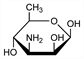
3-Amino-3,6-dideoxymannose; 3-amino-3-deoxyrhamnose; 3,6-dideoxy-3-amino-d-mannopyranose; 3-amino-3,6-dideoxy-d-aldohexose. C6H13NO4; mol wt 163.17.
C 44.17%, H 8.03%, N 8.58%, O 39.22%.
Description and references
An amino sugar which represents the nitrogen-contg moiety of the antifungal antibiotics, nystatin and amphotericin B, q.q.v. Isoln and structure: Dutcher et al., Antibiot. Annu. 1956-1957, 866. Walters et al., J. Am. Chem. Soc. 79, 5076 (1957); Dutcher et al., J. Org. Chem. 28, 995 (1963). Stereochemistry: von Saltza et al., J. Am. Chem. Soc. 83, 2875 (1961); eidem, J. Org. Chem. 28, 999 (1963).
Derivative
N-Acetylmycosamine.C8H15NO5; mol wt 205.21.
C 46.82%, H 7.37%, N 6.83%, O 38.98%.
Properties
Needles from methanol + acetone, mp 195-197°. [α]D22 -46° (ethanol).Derivative
Triacetylmycosamine.C12H19NO7; mol wt 289.28.
C 49.82%, H 6.62%, N 4.84%, O 38.72%.
Properties
Prisms from acetone or ethanol, mp 185-187°. [α]D22 +85° (ethanol).Derivative
Tetraacetylmycosamine.C14H21NO8; mol wt 331.32.
C 50.75%, H 6.39%, N 4.23%, O 38.63%.
Properties
Needles from benzene, mp 159-161°. [α]D23 +39.3° (ethanol).Derivative
Hydrochloride.C6H13NO4.HCl; mol wt 199.63.
C 36.10%, H 7.07%, N 7.02%, O 32.06%, Cl 17.76%.