6360. Nalmefene
Nomenclature
Description and references
Structural analog of naltrexone, q.v., with opiate antagonist activity. Prepn: J. Fishman, US 3814768 (1974 to M. J. Lewenstein); E. F. Hahn et al., J. Med. Chem. 18, 259 (1975). Improved synthesis: P. C. Meltzer, J. W. Coe, EP 140367; eidem, US 4535157 (both 1985 to Key Pharm.). Comparative evaluation of efficacy in mice: R. D. Heilman et al., Res. Commun. Chem. Pathol. Pharmacol. 13, 635 (1976). HPLC determn in human plasma: J. Hsiao, R. Dixon, ibid. 42, 449 (1983). Receptor binding activity in rat brain: M. E. Michel et al., Methods Find. Exp. Clin. Pharmacol. 7, 175 (1985). Clinical evaluation of narcotic antagonism: T. J. Gal, C. A. DiFazio, Anesthesiology 64, 175 (1986). Pharmacokinetics in humans: R. Dixon et al., Clin. Pharmacol. Ther. 39, 49 (1986); eidem, J. Clin. Pharmacol. 27, 233 (1987). Use as antagonist to opioid-induced immobilization in large animals: T. J. Kreeger et al., J. Wildl. Dis. 23, 619 (1987). Comprehensive description: H. G. Brittain, Anal. Profiles Drug Subs. Excip. 24, 351-395 (1996).
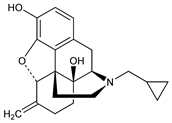
Properties
Crystals from ethyl acetate, mp 188-190°.Derivative
Hydrochloride.Nomenclature
Properties
White to off-white crystalline powder, mp 180-185°. Soly (mg/ml): water (pH 2.25, 5.71, 6.15, 6.25, 7.85, 8.5, 9.15, 10.4): 128; 131; 133; 124; 1.09; 0.18; 0.09; 0.23; methanol 319; ethanol 86.2; acetonitrile 1.07; acetone 0.23; chloroform 0.13. log P (octanol/water): -1.125. pKa 7.63. uv max: 211, 230 nm (am 1977, 778 litre-mol/cm).Therapeutic Category
Narcotic antagonist.
Therapeutic Category (Veterinary)
Narcotic antagonist.Keywords
Narcotic Antagonist