6361. Nalorphine
Nomenclature
CAS number: 62-67-9
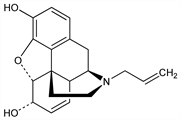
(5α,6α)-7,8-Didehydro-4,5-epoxy-17-(2-propenyl)morphinan-3,6-diol; N-allylnormorphine; allorphine; antorphine; NANM. C19H21NO3; mol wt 311.37.
C 73.29%, H 6.80%, N 4.50%, O 15.42%.
Description and references
Prepd from normorphine: McCawley et al., J. Am. Chem. Soc. 63, 314 (1941); amended procedure: Hart, McCawley, J. Pharmacol. 82, 339 (1944); Weijlard, Erickson, J. Am. Chem. Soc. 64, 869 (1942); US 2364833 (1944); Weijlard, US 2891954 (1959 to Merck & Co.). Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Comprehensive description: M. U. Zubair et al., Anal. Profiles Drug Subs. 18, 195-219 (1989).
Properties
Crystals from ether, mp 208-209° (not 92-93° as first given by McCawley). [α]D25 -155.3° (c = 3 in methanol). Sparingly sol in water, ether. Sol in alc, acetone, chloroform, dil alkalies. uv max (in acid): 285 nm; (in alkali): 298 nm.Derivative
Hydrobromide.Nomenclature
CAS number: 1041-90-3
Lethidrone (Burroughs Wellcome); Norfin (Lusofarmaco). C19H21NO3.HBr; mol wt 392.29.
C 58.17%, H 5.65%, N 3.57%, O 12.24%, Br 20.37%.
Properties
Crystals from alc, dec 258-259°. Sol in water.Derivative
Hydrochloride.Nomenclature
CAS number: 57-29-4
Nalline (Merck & Co.). C19H21NO3.HCl; mol wt 347.84.
C 65.61%, H 6.37%, N 4.03%, O 13.80%, Cl 10.19%.
Properties
Crystals from alc, mp 260-263°. Sol in water. Moderately sol in alc. uv max (water): 285 nm. IR and polarographic data: Seagers et al., J. Am. Pharm. Assoc. Sci. Ed. 41, 640 (1952). pH of 0.5% aq soln: 5.0. LD50 s.c. in rats: 1460 mg/kg (Goldenthal).Note
This is a controlled substance: 21 CFR, 1308.13.Therapeutic Category
Narcotic antagonist.
Therapeutic Category (Veterinary)
Narcotic antagonist.Keywords
Narcotic Antagonist