6524. Nicotine
Nomenclature
Description and references
From the dried leaves of Nicotiana tabacum and N. rustica where it occurs to the extent of 2 to 8%, combined with citric and malic acids. Extraction procedure: Gattermann, Wieland, Laboratory Methods of Organic Chemistry (New York, 24th ed., 1937); Schwyzer, Die Fabrikation pharmazeutischer und chemisch-technischer Produkte (Berlin, 1931). Purification: Ratz, Monatsh. Chem. 26, 1241 (1905). Structure and synthesis: Pinner, Ber. 26, 294 (1893); Pictet, Rotschy, Ber. 37, 1225 (1904); Craig, J. Am. Chem. Soc. 55, 2854 (1933); M. Nakane, C. R. Hutchinson, J. Org. Chem. 43, 3922 (1978). Conformation in soln: T. P. Pitner et al., J. Am. Chem. Soc. 100, 246 (1978). HPLC determn in plasma: M. Harlharan et al., Clin. Chem. 34, 724 (1988). Toxicity data: R. B. Barlow, L. J. McLeod, Br. J. Pharmacol. 35, 161 (1969). Review and bibliography: Jackson, Chem. Rev. 29, 123 (1941). Review of pharmacology: R. W. Ryall in Neuropoisons: Their Pathophysiological Actions vol. 2, L. L. Simpson, D. R. Curtis, Eds. (Plenum, New York, 1974) pp 61-97; N. L. Benowitz, Annu. Rev. Med. 37, 21-32 (1986). Clinical trial of long-term efficacy in smoking cessation: T. Blondal et al., Br. Med. J. 318, 285 (1999). Review of clinical experience in Tourette syndrome: A. A. Silver et al., CNS Spectr. 4, 68-76 (1999).
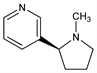
Properties
Colorless to pale yellow, oily liq; very hygroscopic; turns brown on exposure to air or light. Acrid burning taste. Develops odor of pyridine. bp745 247° (partial decompn); bp17 123-125°. Volatile with steam. nD20 1.5282. d420 1.00925. [α]D20 -169.3° (neat); [α]5461 -204.1°. pK1 (15°) 6.16; pK2 10.96. pH of 0.05M soln: 10.2. Forms salts with almost any acid and double salts with many metals and acids. Absorption spectrum: Purvis, J. Chem. Soc. 97, 1035 (1910); Dobbie, Fox, ibid. 103, 1194 (1913). Optical rotatory properties: T. M. Lowry, W. V. Lloyd, J. Chem. Soc. 1929, 1771. Poisonous. Misc with water below 60°; on mixing nicotine with water the volume contracts. Very sol in alc, chloroform, ether, petr ether, kerosene, oils. Distribution of nicotine between water and petroleum oils: Norton, Ind. Eng. Chem. Ind. Ed. 32, 241 (1940). LD50 in mice (mg/kg): 0.3 i.v.; 9.5 i.p.; 230 orally (Barlow, McLeod).Derivative
Hydrochloride.Nomenclature
Properties
Deliquesc crystals. [α]D20 +104° (p = 10).Derivative
Dihydrochloride.Nomenclature
Properties
Deliquesc crystals, very sol in water and alcohol. Nearly insol in ether.Derivative
Sulfate.Nomenclature
Properties
Six-sided tablets. [α]D20 +88° (p = 70). Sol in water, alcohol.Derivative
Bitartrate.Nomenclature
Properties
Dihydrate, crystals. mp 90°. [α]D20 +26° (c = 10). Very sol in water or alcohol.Derivative
Zinc chloride double salt monohydrate.Properties
Also with 4H2O. Very sol in water; sparingly sol in abs alcohol and ether.Derivative
Salicylate.Nomenclature
Properties
Six-sided plates, mp 118°. [α]D20 +13° (c = 9). Freely sol in water or alcohol.Derivative
Polacrilex.Nomenclature
Description and references
Prepn: S. Lichtneckert et al., DE 2136119; eidem, US 3901248 (1972, 1975 both to AB Leo). Review of efficacy in smoking cessation: K. O. Fagerstr"om, Prog. Clin. Biol. Res. 261, 109-128 (1988).
Caution
Nicotine can be absorbed through the alimentary canal, respiratory tract and intact skin. Potential symptoms of overexposure are nausea, salivation, abdominal pain, vomiting, diarrhea; headache, dizziness, auditory and visual disturbances; confusion, weakness, incoordination; paroxysmal atrial fibrillation; convulsions, dyspnea. Death may result from paralysis of respiratory muscles. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 224; Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984) Section III, pp 311-314.Use
Insecticide; fumigant. In the U.S. a 40% soln of nicotine sulfate, Black Leaf 40, was the commonly used form. As a contact poison it is most effective as soap, i.e., as the laurate, oleate, or naphthenate. As a stomach poison a combination with bentonite has come into use.Therapeutic Category
Treatment of smoking withdrawal syndrome.