6525. Nicotinic Acid
Nomenclature
Description and references
Precursor of the coenzymes NAD and NADP, q.q.v. Widely distributed in nature; appreciable amounts are found in liver, fish, yeast and cereal grains. Dietary deficiency is associated with pellagra. The term “niacin” has also been applied to nicotinamide, q.v., or to other derivatives exhibiting the biological activity of nicotinic acid. Prepn by oxidation of alkyl β-substituted pyridines: A. Ladenburg, Ann. 301, 152 (1898). Synthesis from pyridine: S. M. McElvain, M. A. Goese, J. Am. Chem. Soc. 63, 2283 (1941). Prepn by oxidation of nicotine: S. M. McElvain, Org. Synth. coll. vol. I, 385 (1941); C. F. Woodward et al., Ind. Eng. Chem. 36, 540, 544 (1944). Toxicity study: F. G. Brazda, R. A. Coulson, Proc. Soc. Exp. Biol. Med. 62, 19 (1946). LC determn in foods: G. W. Chase, Jr. et al., J. AOAC Int. 76, 390 (1993). Lipid-modifying effect in diabetic patients: M. B. Elam et al., J. Am. Med. Assoc. 284, 1263 (2000). Review of nutritional requirements, bioavailability and relationship with tryptophan: W. J. Darby et al., Nutr. Rev. 33, 289-297 (1975); of biosynthesis, metabolism and physiological effects: L. M. Henderson, Annu. Rev. Nutr. 3, 289-307 (1983); of pharmacology and therapeutic uses: J. R. DiPalma, W. S. Thayer, ibid. 11, 169-187 (1991). Review: “Niacin: Nicotinic Acid, Nicotinamide, NAD(P)” in Vitamins, W. Friedrich, Ed. (Walter de Gruyter, Berlin, 1988) pp 473-542.
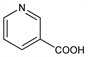
Properties
Needles from alcohol or water, mp 236.6°. Nonhygroscopic and stable in air. Sublimes without decompn. pKa 4.85. pH of satd aq soln 2.7. uv max: 263 nm, Hünecke, Ber. 60, 1451 (1927). One gram dissolves in 60 ml water. Freely sol in boiling water and in boiling alcohol, in alkali hydroxides and carbonates; sol in propylene glycol. Insol in ether. LD50 s.c. in rats: 5 g/kg (Brazda, Coulson).Derivative
N-Oxide see Oxiniacic Acid.Derivative
Sodium salt sesquihydrate.Nomenclature
Direktan (Gerot).Properties
White crystals or crystalline powder; stable in air. One gram dissolves in ≈1.4 ml water, in ≈60 ml alc, in 10 ml glycerol. Insol in ether. pH of aq soln: ≈7.Therapeutic Category
Antilipemic; vitamin (enzyme cofactor).
Therapeutic Category (Veterinary)
Vitamin (enzyme cofactor).Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B3 (Niacin)