6523. Nicotinamide
Nomenclature
Description and references
Precursor of the coenzymes NAD and NADP, q.q.v. The terms niacin, Vitamin B3, and Vitamin PP have been used to refer to both nicotinamide and nicotinic acid, q.v. Isolation: H. v. Euler et al., Z. Physiol. Chem. 258, 212 (1939); P. Karrer, H. Keller, Helv. Chim. Acta 22, 1292 (1939). Prepn from 3-cyanopyridine: E. J. Gasson, D. J. Hadley, US 2904552 (1959 to Distillers). Alternately prepd by passing NH3 gas into molten nicotinic acid: A. Truchan, J. B. Davidson, US 2993051 (1961 to Cowles Chem.). Isoln and analysis by liquid chromatography: C. Bernofsky, Methods Enzymol. 66, 23 (1980). Extraction from cereal products: R. B. Roy, J. J. Merten, J. Assoc. Off. Anal. Chem. 66, 291 (1983). Toxicity data: F. G. Brazda, R. A. Coulson, Proc. Soc. Exp. Biol. Med. 62, 19 (1946). Clinical trial in acne: A. R. Shalita et al., Int. J. Dermatol. 34, 434 (1995); to preserve beta cell function in diabetes: P. Pozzilli et al., Diabetologia 38, 848 (1995). Review: “Niacin: Nicotinic Acid, Nicotinamide, NAD(P)” in Vitamins, W. Friedrich, Ed. (Walter de Gruyter, Berlin, 1988) pp 473-542. Comprehensive description: E. M. Abdel Moety et al., Anal. Profiles Drug Subs. 20, 475-555 (1991).
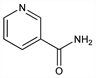
Properties
Needles from benzene, mp 128-131°. Distills at 150-160° at 5×10-4 mm Hg. Absorption spectrum: Kuhn, Vetter, Ber. 68, 2374 (1935). uv max: 261 nm (A1cm1% 451). pK (20°C): 3.3. One gram dissolves in about one ml water, in about 1.5 ml alcohol, in 10 ml glycerol. A 10% w/v soln in water is neutral to litmus. Forms cryst salts with acids. LD50 s.c. in rats: 1.68 g/kg (Brazda, Coulson).Derivative
Compd with ascorbic acid.Nomenclature
Description and references
Prepn: W. Wenner, J. Org. Chem. 14, 22 (1949).
Properties
Yellow crystals, mp 141-145°. [α]D20 +27.5° (c = 8 in H2O). pH of 5% aq soln ≈4.0. Soly (20°): water 40%; abs ethanol 2.4%; methanol 10%. Sparingly sol in acetone. Practically insol in benzene, ether.Therapeutic Category
Antiacne; vitamin (enzyme cofactor).
Therapeutic Category (Veterinary)
Vitamin (enzyme cofactor).Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B3 (Niacin)