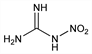
6608. N-Nitroguanidine
Nomenclature
CAS number: 556-88-7
CH4N4O2; mol wt 104.07.
C 11.54%, H 3.87%, N 53.84%, O 30.75%.
Description and references
Exists in two forms. The α-form is the usual stable form. The β-form is converted into the α-form by dissolving in concd H2SO4 and pouring into ice water. Prepn of the α-form from guanidine nitrate and the β-form from guanidine sulfate: T. L. Davis et al., J. Am. Chem. Soc. 47, 1063 (1925). Modified prepn of the α-form: T. L. Davis, Org. Synth. 7, 68 (1927). Absorption spectrum: E. R. Riegel, K. W. Buchwald, J. Am. Chem. Soc. 51, 484 (1929)