6697. Norethindrone
Nomenclature
Description and references
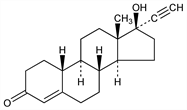
Prepn from 19-nor-4-androstene-3,17-dione: Djerassi et al., J. Am. Chem. Soc. 76, 4092 (1954); US 2744122 (1956 to Syntex); De Ruggieri, US 2849462 (1958). Pharmacokinetics: H. Singh et al., Am. J. Obstet. Gynecol. 135, 409 (1979); M. Humpel, Contraception 26, 83 (1982). Double-blind, comparative clinical trial: S. Koetsawang et al., ibid. 25, 231 (1982); A. Sheth et al., ibid. 243. Multicenter trial of combination with ethinyl estradiol: M. Toews et al., Curr. Ther. Res. 41, 509 (1987). Review of carcinogenicity studies: IARC Monographs 21, 441-460 (1979). Comprehensive description: A. P. Schroff, E. S. Moyer, Anal. Profiles Drug Subs. 4, 268-293 (1975).
Properties
Crystals from ethyl acetate, mp 203-204°. [α]D20 -31.7° (chloroform). First reported as [α]D20 -25° (chloroform). uv max (ethanol): 240 nm (log ε 4.24).Derivative
Mixture with ethinyl estradiol.Nomenclature
Binovum (Ortho-Cilag); Brevicon (Watson); Estrostep (Warner-Lambert); Modicon (Ortho); Neocon 1/35 (Ortho-Cilag); Norimin (Searle); Norinyl 1+35 (Watson); Ortho-Novum (Ortho); Ovcon (Warner-Chilcott); Ovysmen (Janssen-Cilag); Tri-Norinyl (Watson); Trinovum (Janssen-Cilag).Derivative
Mixture with mestranol.Nomenclature
Norinyl-1+50 (Watson); Ortho-Novin 1/50 (Ortho-Cilag); Ortho-Novum 1/50 (Ortho).Derivative
Acetate.Nomenclature
Description and references
Prepn: O. Engelfried et al., US 2964537 (1960 to Schering AG).
Properties
Crystals from methylene chloride + hexane, mp 161-162°. uv max: 240 nm (ε 18690).Derivative
Acetate, mixture with ethinyl estradiol.Nomenclature
Etalontin (Parke-Davis); Primosiston (Schering AG); Anovlar (Schering AG); Gynovlar (Schering AG); Loestrin (Warner-Lambert); Minovlar (Schering AG); Norlestrin (Warner-Lambert).Derivative
Enanthate.Nomenclature
Noristerat (Schering AG).Description and references
Clinical trial as injectable contraceptive: S. K. Banerjee et al., Contraception 30, 561 (1984).
Caution
Norethindrone is reasonably anticipated to be a human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-208.Therapeutic Category
Progestogen. Norethindrone and acetate in combination with estrogen as contraceptive (oral). Enanthate as contraceptive (injectable).
Keywords
Contraceptive (Injectable); Contraceptive (Oral); Progestogen