6825. Oleandomycin
Nomenclature
CAS number: 3922-90-5
PA-105; Amimycin; Landomycin; Romicil. C35H61NO12; mol wt 687.86.
C 61.11%, H 8.94%, N 2.04%, O 27.91%.
Description and references
Antibiotic substance produced by Streptomyces antibioticus no. ATCC 11891: Sobin et al.; Ratajak, Nubel, US 2757123; US 2842481 (1956, 1958 to Pfizer). Structure: Hochstein et al., J. Am. Chem. Soc. 82, 3225 (1960). Absolute configuration: Celmer, ibid. 87, 1797 (1965); Celmer, Hobbs, Carbohydr. Res. 1, 137 (1965); S. Omura et al., Tetrahedron Lett. 1975, 2939. Synthetic study: K. Tatsuta et al., ibid. 29, 3975 (1988). Activity: Hahn, Antibiotics 1, 378, 755 (1967). For a review of macrolide antibiotics see Keller-Schierlein, Fortschr. Chem. Org. Naturst. 30, 313-460 (1973). Toxicity: H. Sous et al., Arzneim.-Forsch. 8, 386 (1958).
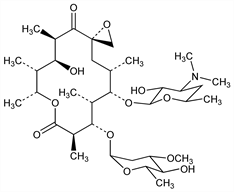
Properties
White amorphous powder. uv max (methanol): 286-289 nm. Moderately sol in water. Sol in dil acids. Freely sol in methanol, ethanol, butanol, acetone. Practically insol in hexane, carbon tetrachloride, dibutyl ether.Derivative
Hydrochloride.Nomenclature
CAS number: 6696-47-5
C35H61NO12.HCl; mol wt 724.32.
C 58.04%, H 8.63%, N 1.93%, O 26.51%, Cl 4.89%.
Properties
Long needles from ethyl acetate, mp 134-135°. [α]D25 -54° (methanol). Freely sol in water. Forms various cryst hydrates. LD50 in mice, rats (mg/kg): 8200, >10000 orally; 600, 400 i.v. (Sous).Derivative
Phosphate.Nomenclature
CAS number: 7060-74-4
Matromycin (Pfizer). C35H61NO12.H3PO4; mol wt 785.85.
C 53.49%, H 8.21%, N 1.78%, O 32.57%, P 3.94%.
Derivative
Triacetyl deriv see Troleandomycin.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Macrolides