6922. Oxantel
Nomenclature
CAS number: 36531-26-7
(E)-3-[2-(1,4,5,6-Tetrahydro-1-methyl-2-pyrimidinyl)ethenyl]phenol; (E)-m-[2-(1,4,5,6-tetrahydro-1-methyl-2-pyrimidinyl)vinyl]phenol; 1-methyl-1,4,5,6-tetrahydro-2-[2-(3-hydroxyphenyl)vinyl]pyrimidine; CP-14445. C13H16N2O; mol wt 216.28.
C 72.19%, H 7.46%, N 12.95%, O 7.40%.
Description and references
Analog of pyrantel, q.v., with activity vs whipworms (Trichuris spp.). Prepn: J. W. Mc Farland, ZA 6804589 corresp to US 3579510 and US 3708584 (1968, 1971, 1973 all to Pfizer). Synthesis and evaluation in whipworm control: J. W. Mc Farland, H. L. Howes J. Med. Chem. 15, 365 (1972). Evaluation vs Trichuris in dogs: H. L. Howes, Proc. Soc. Exp. Biol. Med. 139, 394 (1972). Efficacy vs T. trichiuris in humans: E. L. Lee et al., Am. J. Trop. Med. Hyg. 25, 563 (1976); vs T. suis in swine: M. Robinson, Vet. Parasitol. 5, 223 (1979). Anthelmintic effects of combination with pyrantel pamoate: B. Sinniah, D. Sinniah, Ann. Trop. Med. Parasitol. 75, 315 (1981).
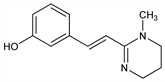
Derivative
Hydrochloride.C13H16N2O.HCl; mol wt 252.74.
C 61.78%, H 6.78%, N 11.08%, O 6.33%, Cl 14.03%.
Properties
Crystals from ethanol, mp 207-208°. uv max (water): 231, 274 nm (ε 12700, 20100).Derivative
Pamoate.Nomenclature
CAS number: 68813-55-8
Oxantel embonate; CP-14445-16; Telopar (Pfizer). C13H16N2O.C23H16O6; mol wt 604.65.
C 71.51%, H 5.33%, N 4.63%, O 18.52%.
Derivative
Mixture with pyrantel pamoate (1:1) see Pyrantel.Therapeutic Category
Anthelmintic (Nematodes).
Therapeutic Category (Veterinary)
Anthelmintic (Nematodes).Keywords
Anthelmintic (Nematodes)