6969. Oxymorphone
Nomenclature
CAS number: 76-41-5
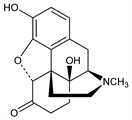
(5α)-4,5-Epoxy-3,14-dihydroxy-17-methylmorphinan-6-one; dihydrohydroxymorphinone; dihydro-14-hydroxymorphinone; 14-hydroxydihydromorphinone. C17H19NO4; mol wt 301.34.
C 67.76%, H 6.36%, N 4.65%, O 21.24%.
Description and references
Semisynthetic opioid analgesic. Prepn from dihydrohydroxycodeinone: U. Weiss, J. Am. Chem. Soc. 77, 5891 (1955); M. J. Lewenstein, U. Weiss, US 2806033 (1957); by demethylation of oxycodone: A. Coop et al., J. Org. Chem. 63, 4392 (1998). Comparative toxicity study: P. E. Tullar, Toxicol. Appl. Pharmacol. 3, 261 (1961). GC-MS determn in blood: R. Meatherall, J. Anal. Toxicol. 29, 301 (2005). Pharmacokinetics of extended release formulation: M. P. Adams, H. Ahdieh, Pharmacotherapy 24, 468 (2004). Review of pharmacology and clinical efficacy: E. Prommer, Support. Care Cancer 14, 109-115 (2006).
Properties
Crystals from boiling ethanol, ethyl acetate or benzene. mp 248-249° (dec). Levorotatory. Sol in boiling acetone and chloroform; readily sol in aq alkalies; moderately sol in boiling ethanol; sparingly sol in benzene. LD50 in mice (mg/g): 0.200 s.c. (Tullar).Derivative
Hydrochloride.Nomenclature
CAS number: 357-07-3
Numorphan (Endo); Opana (Endo). C17H19NO4.HCl; mol wt 337.80.
C 60.44%, H 5.97%, N 4.15%, O 18.95%, Cl 10.50%.
Properties
White or slightly off-white, odorless powder. Sparingly sol in ethanol, ether; freely sol in water. pKa1 8.17; pKa2 9.54. Partition coefficient (octanol/water at 37° and pH 7.4): 0.98Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Therapeutic Category (Veterinary)
Analgesic.Keywords
Analgesic; Opioids; Phenanthrenes