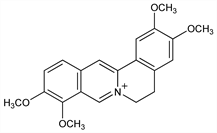
6994. Palmatine
Nomenclature
Description and references
Obtained only in form of its salts. First isolated from Calumba root (Jateorhiza palmata (DC.) Miers, Menispermaceae). Now found in many other genera. Palmatine and tetrahydropalmatine, q.v. are probably the most widely distributed Berberis alkaloids. Extraction procedure: Feist, Dschu, Arch. Pharm. 263, 301 (1925). Structure: Feist, Sandstede, ibid. 256, 2, 5 (1918); Sp"ath, Lang, Ber. 54, 3064, 3068 (1921); Sp"ath, B"ohm, Ber. 55, 2988 (1922); Sp"ath, Meinhard, Ber. 75, 400 (1942). Synthesis: Sp"ath, Quientensky, Ber. 58, 2267 (1925); R. D. Haworth et al., J. Chem. Soc. 1927, 548; Z. Kiparissides et al., Can. J. Chem. 58, 2770 (1980). Identity with calystigine, alkaloid of the Chinese drug Chi-Kuo-Lan: Huang, Chen, C.A. 52, 15827i (1958).