9215. Tetrahydropalmatine
Nomenclature
CAS number: 10097-84-4
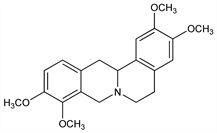
5,8,13,13a-Tetrahydro-2,3,9,10-tetramethoxy-6H-dibenzo[a,g]quinolizine; 2,3,9,10-tetramethoxyberbine; 2,3,9,10-tetramethoxydibenzo[a,g]quinolizidine; hyndarin. C21H25NO4; mol wt 355.43.
C 70.96%, H 7.09%, N 3.94%, O 18.01%.
Description and references
Synthesis of dl-form: Haworth et al., J. Chem. Soc. 1927, 548; Bradsher, Dutta, J. Org. Chem. 26, 2231 (1961); T. Kametani, M. Ihara, J. Chem. Soc. C 1967, 530; G. D. Pandey, K. P.Tiwari, Indian J. Chem. 18B, 545 (1979); Z. Kiparissides et al., Can. J. Chem. 58, 2770 (1980); N. S. Narasimhan et al., Tetrahedron Lett. 22, 2797 (1981). Biosynthesis: D. S. Bhakuni et al., Tetrahedron 36, 2491 (1980). See also Palmatine. Both optically active forms are found in plants.
Derivative
l-Form.Nomenclature
Caseanine; gindarine; rotundine.Properties
Crystals from dil methanol, mp 147°. [α]D20 -291° (c = 0.8 in 95% alcohol). Also a hydrate, mp 115° (effervescence).Derivative
Hydrochloride.C21H25NO4.HCl; mol wt 391.89.
C 64.36%, H 6.69%, N 3.57%, O 16.33%, Cl 9.05%.