7054. Pavoninins
Description and references
Shark repellent compounds isolated from the defense secretion of the Pacific sole, Pardachirus pavoninus. Series of ichthyotoxic and hemolytic steroid amine glycosides; pavoninin-5 is the most abundant. Isoln, structure determn and evaluation of pavoninins 1-6 as shark repellents: K. Tachibana et al., Science 226, 703 (1984); and 13C- and 1H-NMR studies: eidem, Tetrahedron 41, 1027 (1985). Synthesis of pavoninin-1: Y. Ohnishi, K. Tachibana, Bioorg. Med. Chem. 5, 2251 (1997); of pavoninin-4: J. R. Williams et al., J. Org. Chem. 70, 10732 (2005).
Derivative
Pavoninin-1.Nomenclature
CAS number: 94426-01-4
(7α,25R)-7-[[2-(Acetylamino)-2-deoxy-β-d-glucopyranosyl]oxy]-26-(acetyloxy)cholest-4-en-3-one. C37H59NO9; mol wt 661.87.
C 67.14%, H 8.98%, N 2.12%, O 21.76%.
Properties
[α]D20 +19° (c = 1.1 in chloroform). uv max (methanol): 244 nm (Log ε 4.1)Derivative
Pavoninin-4.Nomenclature
CAS number: 94359-66-7
(3α,5α,15α,25R)-26-(Acetyloxy)-3-hydroxycholestan-15-yl
2-(acetylamino)-2-deoxy-β-d-glucopyranoside. C37H63NO9; mol wt 665.90.
C 66.74%, H 9.54%, N 2.10%, O 21.62%.
Properties
White solid, mp 134-136°. [α]D20 +28° (c = 0.4 in ethanol).Derivative
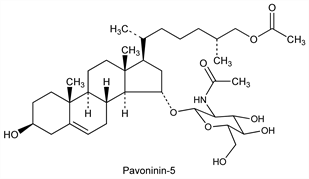
Pavoninin-5.Nomenclature
CAS number: 94480-49-6
(3β,15α,25R)-26-(Acetyloxy)-3-hydroxycholest-5-en-15-yl
2-(acetylamino)-2-deoxy-β-d-glucopyranoside. C37H61NO9; mol wt 663.88.
C 66.94%, H 9.26%, N 2.11%, O 21.69%.