Nomenclature
CAS number: 92-84-2
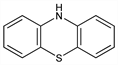
10
H-Phenothiazine; thiodiphenylamine; dibenzothiazine; AFI-Tiazin; Antiverm; Fentiazin; Helmetina; Lethelmin; Nemazine; Orimon; Phénégic; Phenoverm; Phenovis; Phenoxur; Reconox; Souframine; Vermitin.
C
12H
9NS; mol wt 199.27.
C 72.33%, H 4.55%, N 7.03%, S 16.09%.
Description and references
Prepd by fusing diphenylamine with sulfur:
Bernthsen, Ber. 16, 2896 (1883); Ann. 230, 73 (1885); DE 25150 (1883), Frdl. 1, 252. Improved yields
with iodine as catalyst: Knoevenagel, J.
Prakt. Chem. [2] 89, 11 (1914);
Mitchell, Webb, US 2415363 (1947 to Koppers). Purification: Vierling, US 2887482 (1959); Rigby, US 3000887 (1961 to Shell Oil).
Crystal structure: J. D. Bell et al., Chem. Commun. 1968, 1656.
Properties
Yellow, rhombic leaflets or diamond-shaped plates
from toluene or butanol, mp 185.1°. Sublimes at 130° at 1 mm. bp760 371°; bp40 290°. Freely sol in benzene; sol
in ether, in hot acetic acid; slightly sol in alcohol and in mineral
oils. Practically insol in petr ether, chloroform, water. Readily
oxidized by sunlight or when in presence of a finely divided inert
carrier, acquiring a greenish-brown tint. This can be prevented by
the admixture of 0.3-1.0% methenamine.Caution
Potential symptoms of overexposure
are itching, irritation and reddening skin; hepatitis, hemolytic anemia,
abdominal cramps, tachycardia; kidney damage; skin sensitization and
photophobia. See NIOSH Pocket
Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p
248.Use
Insecticide; manuf pharmaceuticals.
Therapeutic Category (Veterinary)
Anthelmintic.
