7330. Pholedrine
Nomenclature
CAS number: 370-14-9
4-[2-(Methylamino)propyl]phenol; p-hydroxy-N,α-dimethylphenethylamine; β-(p-hydroxyphenyl)isopropylmethylamine; α-(p-hydroxyphenyl)-β-methylaminopropane; p-hydroxy-N-methylbenzedrine; Knoll H75. C10H15NO; mol wt 165.23.
C 72.69%, H 9.15%, N 8.48%, O 9.68%.
Description and references
α-Adrenergic agonist. Prepd from p-methoxybenzyl methyl ketone: FR 822422, GB 482414, DE 674753 (1937, 1938 and 1939 to Knoll); Hildebrandt and Hildebrandt, Freese, DE 665793 and DE 767161 (1938 and 1951 to Knoll). Synthesis: Savitskii, Makhnenko, J. Gen. Chem. USSR 10, 1819 (1940); Buzas, Dufour, Bull. Soc. Chim. Fr. 1950, 139. Toxicity data: Lindner, Arch. Exp. Pathol. Pharmakol. 188, 675 (1938).
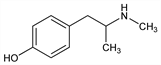
Properties
Crystals from methanol, mp 162-163°. Acrid, burning taste. Alkaline reactions. Slightly sol in water. Sol in alcohol, ether; readily sol in dil acids.Derivative
Sulfate.Nomenclature
CAS number: 6114-26-7
Paredrinol (SK & F); Pulsotyl (Chinoin); Veritol (Knoll). (C10H15NO)2.H2SO4; mol wt 428.54.
C 56.05%, H 7.53%, N 6.54%, O 22.40%, S 7.48%.
Properties
Crystals, dec 320-323°. Sol in water. LD s.c. in rats: 500 mg/kg (Lindner).Therapeutic Category
Antihypotensive.
Therapeutic Category (Veterinary)
Circulatory stimulant.Keywords
α-Adrenergic Agonist; Antihypotensive