7380. Phylloquinone
Nomenclature
Description and references
Photosynthetic electron carrier; occurs widely in green plants, algae, photosynthetic bacteria. Major dietary source of vitamin K, q.v. Isoln from alfalfa: H. Dam et al., Helv. Chim. Acta 22, 310 (1939). Structure: D. W. MacCorquodale et al., J. Biol. Chem. 131, 357 (1939); L. F. Fieser, J. Am. Chem. Soc. 61, 3467 (1939). Partial syntheses from menadione and phytol: H. J. Almquist, A. A. Klose, ibid. 2557; S. B. Binkley et al., ibid. 2558; L. F. Fieser, ibid. 2559. Stereochemistry and total synthesis: H. Mayer et al., Helv. Chim. Acta 47, 221 (1964); L. M. Jackman et al., ibid. 48, 1332 (1965). Synthesis using a π-allylic nickel(I) complex: Sato et al., J. Chem. Soc. Perkin Trans. 1 1973, 2289. Alternate synthesis: Y. Tachibana, Chem. Lett. 1977, 901. Metabolic studies: M. J. Shearer et al., Br. J. Haematol. 18, 297 (1970); 22, 579 (1972). The cis isomer is not bioactive: J. T. Matschiner et al., J. Nutr. 102, 625 (1972). Isoln from chloroplasts: E. Interschick-Niebler, H. K. Lichtenthaler, Z. Naturforsch. 36C 276 (1981). Role in photosystem I: K. Brettel et al., FEBS Lett. 203, 220 (1986). Conversion in vivo to menaquinone-4, q.v.: H. H. W. Thijssen, M. J. Drittij-Reijnders, Br. J. Nutr. 72, 415 (1994). HPLC determn in foods: S. L. Booth et al., J. Agric. Food Chem. 42, 295 (1994). Clinical efficacy in hemorrhagic disease of newborn: P. M. Loughnan, P. N. McDougall, J. Paediatr. Child Health 29, 171 (1993). Comprehensive description: M. M. A. Hassan et al., Anal. Profiles Drug Subs. 17, 449-531 (1988).
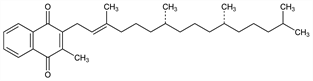
Properties
Yellow viscous oil. [α]D25 -0.28° (dioxane). nD20 1.5263. uv max (petr ether): 242, 248, 260, 269, 325 nm (E1%1cm 396, 419, 383, 387, 68). Insol in water. Sparingly sol in methanol; sol in ethanol, acetone, benzene, petr ether, hexane, dioxane, chloroform, ether, other fat solvents and in vegetable oils. Stable to air and moisture, but dec in sunlight. Unaffected by dil acids, but destroyed by solns of alkali hydroxides and by reducing agents. Keep well closed and protected from light.Derivative
Dihydro form.Nomenclature
Properties
Waxy mass. Freely sol in ether; sparingly sol in petr ether. Insol in water.Derivative
Dihydro form sodium diphosphate.Nomenclature
Properties
mp 138°. Sol in water and methanol.Derivative
2,3-Epoxide.Nomenclature
Description and references
Prepn: Fieser et al., J. Am. Chem. Soc. 61, 3216 (1939).
Properties
Colorless oil. uv max (95% alc): 259, 305 nm (log EM 3.79, 3.31). Insol in water.Therapeutic Category
Vitamin (prothrombogenic).
Therapeutic Category (Veterinary)
Vitamin (prothrombogenic); antidote for dicoumarol poisoning.Keywords
Vitamin/Vitamin Source; Vitamin K