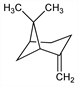
7446. β-Pinene
Nomenclature
Description and references
Found in most essential oils which contain α-pinene, but in far smaller proportions; the l-form occurs most commonly. Initial identification: A. von Baeyer, Ber. 29, 25 (1896). Isoln of the d-form from Ferula galbaniflua Boiss. et Buhse, Umbelliferae: B. N. Rutovski, I. V. Vinogradova, J. Prakt. Chem. 120, 41 (1928); from Cynomarathrum nuttallii A. Gray, Umbelliferae: E. K. Nelson, J. Am. Chem. Soc. 55, 3400 (1933). Irreversible isomerization of β-pinene to α-pinene occurs on shaking with platinum black satd with hydrogen: F. Richter, W. Wolff, Ber. 59, 1733 (1926). Synthesis: G. Bonnet, Bull. Inst. Pin 1938, 217; 1939, 1, C.A. 33, 42234 (1939); K. J. Crowley, Proc. Chem. Soc. London 1962, 245; Tetrahedron 21, 1001 (1965); L. M. Harwood, M. Julia, Synthesis 1980, 456. For general refs see α-pinene.