7458. Pipecolic Acid
Nomenclature
CAS number: 535-75-1
2-Piperidinecarboxylic acid; pipecolinic acid; hexahydropicolinic acid; homoproline; dihydrobaikiaine. C6H11NO2; mol wt 129.16.
C 55.79%, H 8.58%, N 10.84%, O 24.77%.
Description and references
The l-form occurs in plants: Phillips, Chem. Ind. (London) 1953, 127. Prepn: A. Ladenburg, Ber. 24, 640 (1891); Stevens, Ellman, J. Biol. Chem. 182, 75 (1950); V. Asher et al., Tetrahedron Lett. 22, 141 (1981). Synthesis of l-pipecolic acid from l-lysine: Fujii, Miyoshi, Bull. Chem. Soc. Jpn. 48, 1341 (1975). Synthesis of racemate: R. T. Shuman et al., J. Org. Chem. 55, 738 (1990).
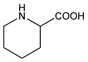
Derivative
l-Form.Properties
Needles by sublimation, mp 270°. [α]D25 -34.9°. Sol in water, dil alcohol. Sparingly sol in abs alcohol, acetone, chloroform. Insol in ether.Derivative
d-Form.Properties
Platelets from alcohol, mp 270°. [α]D25 +35.7°. Sol in water. Somewhat sol in alcohol.Derivative
dl-Form.Properties
Leaflets from water, mp 264°. Sol in water, boiling alcohol.Derivative
dl-Hydrochloride.C6H11NO2.HCl; mol wt 165.62.
C 43.51%, H 7.30%, N 8.46%, O 19.32%, Cl 21.41%.