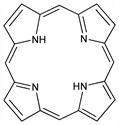
7599. Porphine
Nomenclature
Description and references
Parent substance of the porphyrins, a group of compounds found in all living matter which are the basis of respiratory pigments in animals and plants. In porphyrins, side chains are substituted for the hydrogens in the porphine pyrrole rings. Chlorins are dihydroporphyrins. See also: chlorophyll, hemoglobin, vitamin B12, hematin. Prepn of porphine: Fischer, Gleim, Ann. 521, 157 (1935); Rothemund, J. Am. Chem. Soc. 58, 625 (1936); Krol, J. Org. Chem. 24, 2065 (1959). Study of porphyrin analogs: C. L. Honeybourne et al., Tetrahedron 36, 1833 (1980). Review of biosyntheses of porphyrins and chlorins: A. R. Battersby, E. McDonald in Porphyrins Metalloporphyrins, K. M. Smith, Ed. (Elsevier, New York, 1975) pp 61-122. Review of porphyrin syntheses: R. P. Evstigneeva, Pure Appl. Chem. 53, 1129-1140 (1981). Comprehensive seven volume treatise: The Porphyrins, D. Dolphin, Ed. (Academic Press, New York, 1978).