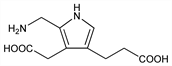
7600. Porphobilinogen
Nomenclature
CAS number: 487-90-1
5-(Aminomethyl)-4-(carboxymethyl)-1H-pyrrole-3-propanoic
acid; 2-aminomethylpyrrol-3-acetic acid 4-propionic acid. C10H14N2O4; mol wt 226.23.
C 53.09%, H 6.24%, N 12.38%, O 28.29%.
Description and references
An intermediate in the biosynthesis of heme, q.v., found in the urine of patients with acute porphyria: Westall, Nature 170, 614 (1952). Isoln and structure: Cookson, Rimington, Biochem. J. 57, 476 (1954). Synthesis: Jackson, MacDonald, Can. J. Chem. 35, 715 (1957). Chemistry of conversion into porphyrins: Mathewson, Corwin, J. Am. Chem. Soc. 83, 135 (1961).