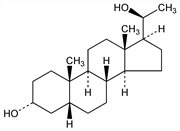
7727. Pregnanediol
Nomenclature
Description and references
A metabolite of progesterone, present in large amounts in pregnancy urine. Isoln from pregnancy urine of women: Marrian, Biochem. J. 23, 1090 (1929), Butenandt, Ber. 63, 659 (1930); of cows, mares, and chimpanzees: Fish et al., J. Biol. Chem. 143, 716 (1942). Prepn by reduction of pregn-16-ene-3,20-dione: Marker et al., US 2352852 (1944 to Parke, Davis). Conversion to progesterone: Butenandt, Schmidt, Ber. 67, 1893, 1901 (1934). Conversion to 3α-hydroxypregnan-20-one: Marker, US 2223377 (1940 to Parke, Davis). Prepn of the 3-acetate: Hirschmann, J. Biol. Chem. 140, 797 (1941); Ralls et al., ibid. 210, 709 (1954). Prepn of the 20-acetate: Hirschmann, loc. cit. Prepn of the diacetate: Johnson et al., J. Chem. Soc. 1954, 1302. Crystal structure: Haner, Norton, Acta Crystallogr. 16, 707 (1963). Review of metabolism, bioactivity and assay during pregnancy: P. J. Keller, Contrib. Gynecol. Obstet. 2, 75-91 (1976).