7774. Proglumetacin
Nomenclature
CAS number: 57132-53-3
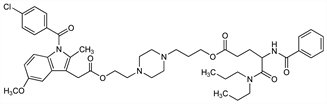
1-(4-Chlorobenzoyl)-5-methoxy-2-methyl-1H-indole-3-acetic
acid 2-[4-[3-[[4-(benzoylamino)-5-(dipropylamino)-1,5-dioxopentyl]oxy]propyl]-1-piperazinyl]ethyl
ester; (±)-N-[2-[1-(p-chlorobenzoyl)-5-methoxy-2-methyl-3-indolylacetoxy]ethyl]-N′-[3-(N-benzoyl-N′,N′-di-n-propyl-dl-isoglutaminoyl)oxypropyl]piperazine. C46H58ClN5O8; mol wt 844.43.
C 65.43%, H 6.92%, Cl 4.20%, N 8.29%, O 15.16%.
Description and references
Deriv of indomethacin, q.v. Prepn: F. Makovec et al., DE 2535799 corresp to US 3985878 (both 1976 to Rotta). Series of articles on pharmacology, mechanism of action, safety: Arzneim.-Forsch. 29, 1116-1129 (1979). Bioavailability study: A. A. Bignamini, P. L. Casula, Curr. Med. Res. Opin. 6, 299 (1979). Clinical evaluation: J. Münzenberg, S. Tachibana, Pharmatherapeutica 2, 279 (1980); P. Loizzi et al., ibid. 285. Mutagenicity study: R. Vidal y Plana et al., Farmaco Ed. Prat. 33, 543 (1978). Toxicity data for dimaleate: A. L. Rovati et al., Arzneim.-Forsch. 29, 1116 (1979).
Derivative
Dimaleate.Nomenclature
CAS number: 59209-40-4
Protacine; CR-604; Afloxan (Rottapharm); Miridacin (Taiho); Protaxon (Opfermann); Proxil (Rottapharm). C46H58ClN5O8.2C4H4O4; mol wt 1076.58.
C 60.24%, H 6.18%, Cl 3.29%, N 6.51%, O 23.78%.
Properties
Crystals from ethanol, mp 146-148°. LD50 in male mice, rats (mg/kg): 262, 450 orally (Rovati).Therapeutic Category
Anti-inflammatory.
Keywords
Anti-inflammatory (Nonsteroidal); Arylacetic Acid Derivatives