7838. Propoxycarbazone
Nomenclature
CAS number: 145026-81-9
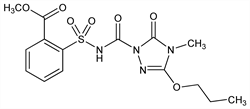
2-[[[(4,5-Dihydro-4-methyl-5-oxo-3-propoxy-1H-1,2,4-triazol-1-yl)carbonyl]amino]sulfonyl]benzoic
acid methyl ester; methyl 2-[[[(4-methyl-5-oxo-3-propoxy-4,5-dihydro-1H-1,2,4-triazol-1-yl)carbonyl]amino]sulfonyl]benzoate. C15H18N4O7S; mol wt 398.39.
C 45.22%, H 4.55%, N 14.06%, O 28.11%, S 8.05%.
Description and references
Triazolinone graminicide for use in cereal crops; acetolactate synthase inhibitor. Prepn: K.-H. Müller et al., EP 507171; eidem, US 5534486 (1992, 1996 both to Bayer). Review of properties and field trials: D. Feucht et al., Brighton Crop Prot. Conf. - Weeds 1999, 53-58. Absorption and fate in weeds: L. Fandrich et al., Weed Sci. 49, 717 (2001).
Properties
mp 141°. pKa: 2.1.Derivative
Sodium salt.Nomenclature
CAS number: 181274-15-7
BAY MKH 6561; Attribut (Bayer CropSci.); Olympus (Bayer CropSci.). C15H17N4NaO7S; mol wt 420.37.
C 42.86%, H 4.08%, N 13.33%, Na 5.47%, O 26.64%, S 7.63%.