7896. Protopine
Nomenclature
CAS number: 130-86-9
4,6,7,14-Tetrahydro-5-methyl-bis[1,3]benzodioxolo[4,5-c:5′,6′-g]azecin-13(5H)-one; 7-methyl-2,3:9,10-bis(methylenedioxy)-7,13a-secoberbin-13a-one; fumarine; macleyine. C20H19NO5; mol wt 353.37.
C 67.98%, H 5.42%, N 3.96%, O 22.64%.
Description and references
From opium: Hesse, Ber. 4, 693 (1871); also from the herb Fumaria officinalis L., Chelidonium majus L., and many other Papaveraceae and Fumariaceae: Manske in The Alkaloids vol. IV, R. H. F. Manske, H. L. Holmes, Eds. (Academic Press, New York, 1954) pp 157-159. Structure: Perkin, Jr., J. Chem. Soc. 109, 815 (1916); Gadamer, Bruchhausen, Arch. Pharm. 260, 97 (1922); Mottus et al., Can. J. Chem. 31, 1144 (1953); Anet, Marion, ibid. 32, 452 (1954). Synthesis: Haworth, Perkin, J. Chem. Soc. 1926, 1769. Crystal structure: Hall, Ahmed, Acta Crystallogr. 24B, 337 (1968).
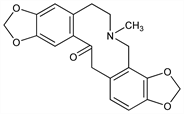
Properties
Monoclinic prisms from alcohol + chloroform, mp 208°. d 1.399 (calc). uv max (95% ethanol): 293 nm (log ε 3.93). Sol in 15 parts chloroform, 900 parts alc, 1000 parts ether. Slightly sol in ethyl acetate, carbon disulfide, benzene, petr ether. Practically insol in water.Derivative
Hydrochloride.C20H19NO5.HCl; mol wt 389.83.
C 61.62%, H 5.17%, N 3.59%, O 20.52%, Cl 9.09%.
Properties
Prisms from alcohol, sol in 143 parts water at 13°, sol in alcohol. Also a hexahydrate, needles from water.Derivative
Methiodide.C20H19NO5.CH3I; mol wt 495.31.
C 50.92%, H 4.48%, N 2.83%, O 16.15%, I 25.62%.