7897. Protoporphyrin IX
Nomenclature
CAS number: 553-12-8
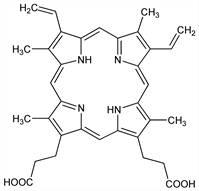
7,12-Diethenyl-3,8,13,17-tetramethyl-21H,23H-porphine-2,18-dipropanoic acid; 3,7,12,17-tetramethyl-8,13-divinyl-2,18-porphinedipropionic
acid; 1,3,5,8-tetramethyl-2,4-divinylporphine-6,7-dipropionic acid; ooporphyrin; Kammerer's porphyrin. C34H34N4O4; mol wt 562.66.
C 72.58%, H 6.09%, N 9.96%, O 11.37%.
Description and references
Biological precursor of blood and plant pigments. Prepd from hemin: Fischer-Orth, Die Chemie des Pyrrols II, 1, 396 (Leipzig, 1937); Ramsey, Biochem. Prep. 3, 39 (1953). Structure: Sparatore, Mauzerall, J. Org. Chem. 25, 1073 (1960). Synthesis: Carr et al., J. Chem. Soc. C 1971, 487. Crystal and molecular structure: W. S. Caughey, J. A. Ibers, J. Am. Chem. Soc. 99, 6639 (1977). Chelates with metals, esp iron, in the ferrous state to form heme, q.v., in the ferric state to form hematin, q.v. Review: Rimington, Kennedy, in M. Florkin, H. S. Mason, Comparative Biochemistry (Academic Press, New York, 1962) pp 557-614.
Properties
Monoclinic, brownish-yellow prisms from ether. Absorption max (25% HCl): 602.4, 582.2, 557.2 nm. Freely sol in chloroform, glacial acetic acid, alcohol contg HCl, ether contg some glacial acetic acid, hydrochloric acid. Somewhat sol in dil alkalies, aniline, pyridine. Forms sparingly sol disodium and dipotassium salts.Derivative
Disodium salt.Nomenclature
CAS number: 50865-01-5
Depocolin-S (Ohta). C34H32N4Na2O4; mol wt 606.62.
C 67.32%, H 5.32%, N 9.24%, Na 7.58%, O 10.55%.
Derivative
Dimethyl ester.Nomenclature
CAS number: 5522-66-7
C36H38N4O4; mol wt 590.71.
C 73.20%, H 6.48%, N 9.48%, O 10.83%.
Properties
Crystals from chloroform + methanol, mp 228-230°. Absorption max (25% HCl): 601, 556, 409 nm. Soluble in chloroform, slightly sol in methanol. Insol in sodium carbonate solns.Therapeutic Category
In liver disease.
Keywords
Hepatoprotectant