Nomenclature
CAS number: 7633-29-6
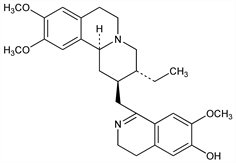
1′,15-Didehydro-7′,10,11-trimethoxyemetan-6′-ol.
C
28H
36N
2O
4; mol wt 464.60.
C 72.38%, H 7.81%, N 6.03%, O 13.77%.
Description and references
Minor alkaloid found in ipecac, the ground
roots of Uragoga ipecacuanha (Brot.) Baill. [Cephaelis
ipecacuanha (Brot.) A. Rich.], Rubiaceae: F. H.
Carr, F. L. Pyman, J. Chem. Soc. 105, 1591 (1914); O. Hesse, Ann. 405, 1 (1914). Structure and stereochemistry: A. R.
Battersby et al., J. Chem. Soc. 1959, 2704, 3512. Mass spectrum: H. Budzikiewicz et al., Tetrahedron 20, 399 (1964); 1H,13C-NMR of O-methylpsychotrine: T. Fujii et al., Chem. Pharm. Bull. 30, 598 (1982); 13C-NMR of psychotrine: eidem, ibid. 31, 2583 (1983). Proposed alternate
structure with exocyclic double bond: E. E. van Tamelen et al., J. Am. Chem. Soc. 79, 4817 (1957); C. Schuij et al., J. Chem. Soc. Perkin Trans. 1 1979, 970. Review: M. Janot in Manske, Holmes, The Alkaloids vol. 3 (Academic Press, New
York, 1953) pp 363-394.
Derivative
Tetrahydrate.
Properties
Yellow prisms with blue fluorescence from dil
acetone or alcohol. Very bitter taste, produces
nausea instantly. The anhydr material sinters at 120°,
becomes transparent at 120-126° and melts completely at 128°. [α]D15 +69.3° (c = 2 in alcohol, calcd as the tetrahydrate). Sparingly
sol in water, benzene, petr ether, ether. More sol in alcohol, acetone,
chloroform. uv max (0.1N HCl): 240, 288, 306, 356 nm (ε 13900, 5700, 6250, 6800).Derivative
Sulfate trihydrate.
C
28H
36N
2O
4.H
2SO
4.3H
2O; mol wt 616.72.
C 54.53%, H 7.19%, N 4.54%, O 28.54%, S 5.20%.
Properties
Pale yellow scales from water, dec 214-217° when
anhydr. [α]D20 +39.2°.Derivative
O-Methylpsychotrine.
Properties
Has a methoxy group in place of the hydroxyl group.
mp 123-124°. [α]D20 +43.2° (alc). uv max (water) pH 1: 241.5, 288.5, 305, 354 (log ε 4.26, 3.86, 3.92, 3.91);
pH 13: 226, 278.5, 307 (log ε 4.43, 3.96, 3.77).