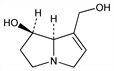
8166. Retronecine
Nomenclature
Description and references
The most common base portion of pyrrolizidine alkaloids. “Necine” bases are 1-methylpyrrolizidines of different stereochemical configurations and degrees of hydroxylation that occur in the form of esters in alkaloids of Senecio, Crotalaria and a number of genera of the Boraginaceae. Retronecine occurs in nature in the (+)-form. It is the necine base of monocrotaline, senecionine, seneciphylline, retrorsine, q.q.v., and numerous other hepatotoxic pyrrolizidine alkaloids. Structure: R. Adams, E. F. Rogers, J. Am. Chem. Soc. 63, 228 (1941). Total synthesis: T. A. Geissman, A. C. Waiss, J. Org. Chem. 27, 139 (1962). Stereospecific synthesis: E. Vedejs, G. R. Martinez, J. Am. Chem. Soc. 102, 7993 (1980); H. Niwa et al., Tetrahedron Lett. 27, 4605 (1986). Total synthesis of (-)-form: J. Cooper et al., Chem. Commun. 1988, 509. Toxicity data: P. N. Harris et al., J. Pharmacol. Exp. Ther. 75, 78 (1942). Biosynthesis: D. J. Robins, J. R. Sweeney, Chem. Commun. 1979, 120; G. Grue Sorensen, I. D. Spenser, J. Am. Chem. Soc. 103, 3208 (1981). Comprehensive reviews on retronecine and other necine bases: F. L. Warren, Fortschr. Chem. Org. Naturst. 12, 198-269 (1955); 24, 329-406 (1966); D. J. Robins, ibid. 41, 115-203 (1982); F. L. Warren in Alkaloids Vol. XII, R. H. F. Manske, Ed. (Academic Press, New York, 1970) pp 245-331.