Nomenclature
CAS number: 81-07-2
1,2-Benzisothiazol-3(2
H)-one 1,1-dioxide; 2,3-dihydro-3-oxobenzisosulfonazole; 1,2-dihydro-2-ketobenzisosulfonazole; saccharin insoluble; benzosulfimide;
o-sulfobenzimide; benzoic sulfimide;
o-sulfobenzoic acid imide; Gluside; Glucid; Garantose; Saccharinol; Saccharinose; Saccharol; Saxin (Burroughs Wellcome); Sykose; Hermesetas.
C
7H
5NO
3S; mol wt 183.18.
C 45.90%, H 2.75%, N 7.65%, O 26.20%, S 17.50%.
Description and references
Prepn from o-sulfamoylbenzoic acid:
Remsen, Fahlberg, Ber. 12, 470 (1879). History and comparison with other sweetening agents:
R. W. Moncrieff, The Chemical Senses (Wiley, New York, 1946). Mfg processes: O. Beyer, Handbuch der Saccharinfabrikation (Rascher,
Zürich, 1923); FIAT Report PB 901 (1945); Chem. Eng. (N.Y.) 61, 128, 150 (July 1954). Maumee Chemical's process:
Chem. Eng. News 41, 76-78 (Dec. 9, 1963). Stability studies: DeGarmo et al., J. Am. Pharm. Assoc. Sci. Ed. 41, 17 (1952). Toxicity data: J. D. Taylor et
al., Food Cosmet. Toxicol. 6, 313 (1968). Series of articles on toxicology and
metabolism: Food Chem. Toxicol. 23, 419-546 (1985). Review of toxicology and carcinogenicity
studies of saccharin and its salts: IARC
Monographs 22, 111-170 (1980); D. L. Arnold, Fundam. Appl. Toxicol. 4, 674-685
(1984). Review: R. Mazur, “Sweeteners” in Kirk-Othmer Encyclopedia of Chemical Technology vol. 22 (John Wiley & Sons, New York, 3rd ed., 1983)
pp 448-464.
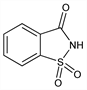
Properties
Monoclinic crystals, mp 228.8-229.7°. Twinning on (001). Perfect
100 cleavage. Acicular crystals by vacuum sublimation. In dil aq
soln it is 500 times as sweet as sugar; the sweet taste is still detectable
in 1:100,000 dilution. Bitter, metallic aftertaste. d 0.828. Heat of combustion at constant
volume: 4753.1 cal/g. uv max (0.1N NaOH): 267.3 nm (ε 1570). One gram dissolves in 290 ml water, 25 ml boiling water, 31 ml
alcohol, 12 ml acetone, about 50 ml glycerol; freely sol in solns
of alkali carbonates; slightly sol in chloroform, ether. pH of 0.35%
aq soln 2.0. Hydrolysis products of saccharin are o-sulfamoylbenzoic
acid (alkaline hydrolysis) and ammonium o-sulfobenzoic
acid (acid hydrolysis).Derivative
Ammonium salt.
Nomenclature
Saccharin ammonium; Daramin; Sucline (Soekami). C
7H
8N
2O
3S; mol wt 200.22.
C 41.99%, H 4.03%, N 13.99%, O 23.97%, S 16.01%.
Derivative
Sodium salt dihydrate.
Nomenclature
Soluble saccharin; saccharin sodium; Kristallose; Crystallose (Tenneco); Dagutan; Sucaryl (Abbott); Sucromat (Mayoly-Spindler). C
7H
4NNaO
3S.2H
2O; mol wt 241.20.
C 34.86%, H 3.34%, N 5.81%, Na 9.53%, O 33.17%, S 13.29%.
Properties
Crystalline powder; effloresces in dry air. In
dil aq soln it is 300-500 times as sweet as sugar. One gram dissolves
in 1.2 ml water, in about 50 ml alcohol. Aq solns are neutral or
alkaline to litmus, but not alkaline to phenolphthalein. LD50 in mice, rats (g/kg): 6.3, 7.1 i.p.; 17.5, 17.0 orally (Taylor). Anhydrous form, Sucrédulcor.Note
Saccharin was formerly listed as reasonably
anticipated to be a human carcinogen: Eighth
Report on Carcinogens (PB99-128746, 1998) p III-905; delisted
because the cancer data are not sufficient to meet the current criteria
for this listing: Ninth Report on Carcinogens (PB2000-107509, 2000) p B-3.Use
Non-nutritive sweetener; pharmaceutic aid (flavor).
In formulations for electroplating-bath brighteners.
