8346. Samandarine
Nomenclature
CAS number: 467-51-6
1α,4α-Epoxy-3-aza-A-homoandrostan-16β-ol. C19H31NO2; mol wt 305.45.
C 74.71%, H 10.23%, N 4.59%, O 10.48%.
Description and references
Constitutes together with samandarone approx 75% of the alkaloid content in the skin glands of the European fire and Alpine salamanders: Salamandra maculosa resp. atra Laur., Salamandridae. Isoln: Sch"opf, Braun, Ann. 514, 69 (1934). Structure and configuration: W"olfel et al., Ber. 94, 2361 (1961).
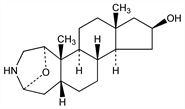
Properties
Needles from abs methanol or 50% acetone, mp 187-188°. Solvated crystals from dil methanol. [α]D17 +43.3° (acetone). Freely sol in most organic solvents. Practically insol in water and NaOH solns.Derivative
Hydrochloride.C19H31NO2.HCl; mol wt 341.92.
C 66.74%, H 9.43%, N 4.10%, O 9.36%, Cl 10.37%.
Properties
Crystals from methanol, mp 312-313°; from water, mp 321-322° (monohydrate).Derivative
Samandarone.C19H29NO2; mol wt 303.44.
C 75.21%, H 9.63%, N 4.62%, O 10.55%.
Description and references
ketone corresponding to the secondary alcohol in samandarine. Synthesis: Hara, Oka, J. Am. Chem. Soc. 89, 1041 (1967).