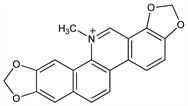
8356. Sanguinarine
Nomenclature
Description and references
Principal alkaloid in sanguinaria extract. Isolated from the root of Sanguinaria canadensis L., and other Papaveraceae: Schmidt et al., Arch. Pharm. 231, 145 (1893). Widely distributed in poppy-fumaria species; constituent of argemone oil: S. A. E. Hakim et al., Nature 189, 198 (1961). Identity with pseudochelerythrine: Gadamer, Stichel, ibid. 262, 488 (1924). Structure: Sp"ath, Kuffner, Ber. 64, 370, 2034 (1931); Beke, Acta Chim. Acad. Sci. Hung. 17, 463 (1958). Biosynthesis: Leete, J. Am. Chem. Soc. 85, 473 (1963). Synthesis: Dyke et al., Tetrahedron Lett. 1968, 3933; Sainsbury et al., J. Chem. Soc. C 1970, 1797; Onda et al., Chem. Pharm. Bull. 17, 404 (1969); 19, 31 (1971); M. Hanaoka et al., Chem. Lett. 22, 739 (1986). Purification procedure: Stipanovic et al., J. Heterocycl. Chem. 9, 1453 (1972). Studies of effect of sanguinarine and argemone oil on intraocular pressure: S. A. E. Hakim, Br. J. Ophthalmol. 38, 193 (1954); G. C. Dobbie, M. E. Langham, ibid. 45, 81 (1961). Anti-inflammatory activity in rats: J. Lenfeld et al., Planta Med. 43, 161 (1981). Clinical study of sanguinarine oral rinse as antiplaque agent: G. L. Southard et al., J. Am. Dent. Assoc. 108, 338 (1984). Acute toxicity study: P. J. Becci et al., J. Toxicol. Environ. Health 20, 199 (1987). Review: M. Shamma, The Isoquinoline Alkaloids (Academic Press, New York, 1972) pp 315-343. Symposium on chemistry, antimicrobial effects, clinical studies and toxicology: J. Can. Dent. Assoc. 56, Suppl 7, 5-47 (1990).