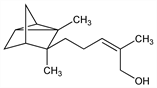
8357. α-Santalol
Nomenclature
Description and references
A sesquiterpene alcohol which together with β-santalol comprises about 90% of commercial sandalwood oil: F. W. Semmler, K. Bode, Ber. 40, 1124 (1907); E. Gruenther, The Essential Oils vol. 2 (Van Nostrand, New York, 1949) pp 266-269. Structure: F. W. Semmler, Ber. 43, 1893 (1910). Separation of α- and β-santalols of high purity: A. E. Bradfield et al., J. Chem. Soc. 1935, 309. Synthesis of α-santalol and conversion to β-santalol: S. C. Bhattacharyya, Sci. Cult. 13, 207 (1947), C.A. 43, 5484gh (1949); see also V. M. Sathe et al., Indian J. Chem. 4, 393 (1966); S. Y. Kamat et al., Tetrahedron 23, 4487 (1967). Stereochemistry studies: G. Brieger, Tetrahedron Lett. 1963, 2123. Natural α-santalol has a cis-(+)-configuration. Total synthesis: R. G. Lewis et al., ibid. 1967, 401. Stereospecific synthesis: E. J. Corey et al., J. Am. Chem. Soc. 92, 226, 6314 (1970); M. Julia, P. Ward, Bull. Soc. Chim. Fr. 1973, 3065; M. Tamura, G. Suzukamo, Tetrahedron Lett. 22, 577 (1981). Synthesis and separation of cis- and trans-isomers: K. Sato et al., Bull. Chem. Soc. Jpn. 49, 3351 (1976). Review: J. Simonsen, D. H. R. Barton, The Terpenes vol. 3 (Univ. Press, Cambridge, 1961) pp 178-188; A. Bhati, Flavour Ind. 1, 235-251 (1970).