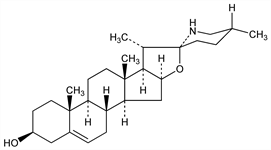
8709. Solasodine
Nomenclature
CAS number: 126-17-0
(3β,22α,25R)-Spirosol-5-en-3-ol; solasod-5-en-3β-ol; Δ5-20βF,22αF,25αF,27-azaspirosten-3β-ol; solancarpidine; solanidine-S; purapuridine. C27H43NO2; mol wt 413.64.
C 78.40%, H 10.48%, N 3.39%, O 7.74%.
Description and references
Steroidal alkaloid isolated from various Solanum species. By hydrolysis of solasonine: Rochelmeyer, Arch. Pharm. 277, 329 (1939). See also ref under Solasonine and Solanidine. Structure: Briggs et al., J. Chem. Soc. 1950, 3013. Synthesis: Uhle, J. Org. Chem. 27, 656 (1962); Schreiber, Rnsch, Tetrahedron 20, 1939 (1964); Kessar et al., ibid. 27, 2869 (1971). Comprehensive description: G. Indrayanto et al., Anal. Profiles Drug Subs. Excip. 24, 487-522 (1996).