9107. Tectorigenin
Nomenclature
Description and references
Phytoalexin aglycon isolated from rhizomes of Iris tectorum Maxim., Iridaceae, the extract of which is used as a traditional Chinese medicine. Isoln of the glycoside and aglycon: S. Shibata, J. Pharm. Soc. Jpn. 37, 380 (1927), C.A. 21, 30508 (1927); from Iris germanica Linnaeus: A. Kawase et al., Agric. Biol. Chem. 37, 145 (1973). Structure: Asahina et al., J. Pharm. Soc. Jpn. 48, 1087 (1928), C.A. 23, 27181 (1929); Shriner et al., J. Am. Chem. Soc. 61, 2322 (1939). Synthesis: Farkas, Várady, Ber. 93, 1269 (1960); W. Baker et al., J. Chem. Soc. C 1970, 1219. Inhibition of cyclooxygenase-2 induction: K. Ohuchi et al., Recent Adv. Nat. Prod. Res., Proc. 3rd Int. Symp. 1999, 12-24. Antioxidant effects: K.-T. Lee et al., Arch. Pharmacal Res. 23, 461 (2000).
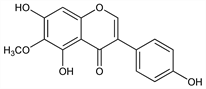
Properties
Pale yellow needles from ethanol, mp 225-226°. uv max (ethanol): 267-269, 338 nm (log ε 4.41, 3.40).Derivative
Tectoridin.Nomenclature
Description and references
Identity with shekanin and structure: Mannich et al., Arch. Pharm. 275, 317 (1937). Synthesis: Várady, Acta Chim. Acad. Sci. Hung. 48, 181 (1966).