9117. Telbivudine
Nomenclature
Description and references
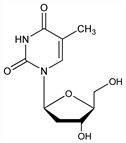
Nucleoside analog; specific inhibitor of hepatitis B virus (HBV) replication. Prepn: J. Smejkal, F. Sorm, Collect. Czech. Chem. Commun. 29, 2809 (1964); A. Holy, ibid. 37, 4072 (1972). Anti-HBV activity: M. L. Bryant et al., Nucleosides Nucleotides Nucleic Acids 20, 597 (2001) DOI PubMed. In vitro pharmacology: B. Hernandez-Santiago et al., Antimicrob. Agents Chemother. 46, 1728 (2002) DOI PubMed. Clinical evaluation in hepatitis B: C.-L. Lai et al., Hepatology 40, 719 (2004) DOI PubMed. Review of mechanism of action and pharmacology: D. N. Standring et al., Antivir. Chem. Chemother. 12, Suppl. 1, 119-129 (2001) PubMed; of clinical development: S.-H. B. Han, Expert Opin. Invest. Drugs 14, 511-519 (2005) DOI. See also Thymidine.
Properties
White to slightly yellowish powder. mp 189° (Holy). Also reported as crystals from ethanol, mp 186° (Smejkal, Sorm). [α]D20 -20.3° (c = 0.192 in water). Soly (mg/ml): water >20; ethanol 0.7; n-octanol 0.1. uv max (pH 2): 267 nm (ε 9800).Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides