9169. Terpin
Nomenclature
CAS number: 80-53-5
4-Hydroxy-α,α,4-trimethylcyclohexanemethanol; p-menthane-1,8-diol; dipenteneglycol. C10H20O2; mol wt 172.26.
C 69.72%, H 11.70%, O 18.58%.
Description and references
Both cis-and trans-modifications are known. The cis-compd is obtained most readily in the hydrated form, cis-terpin hydrate. Prepn of cis-form from oil of turpentine: Hempel, Ann. 180, 71 (1876); Wallach, Ann. 230, 225 (1885); Schmitt, Manuf. Chem. 26, 350 (1955). From d-limonene: Sword, J. Chem. Soc. 127, 1632 (1925). Prepn of trans-form from 1,8-cineole, α-terpineol or cis-terpin hydrate: Matsuura et al., Bull. Chem. Soc. Jpn. 31, 990 (1958); Lombard, Ambroise, Bull. Soc. Chim. Fr. 1961, 230. Structure of cis- and trans-forms: Baeyer, Ber. 26, 2861 (1893); Wagner, ibid. 27, 1636 (1894).
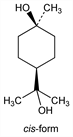
Derivative
cis-Form hydrate.Nomenclature
CAS number: 2451-01-6
Terpin hydrate; terpinol. Properties
Rhombic pyramids from water, mp 116-117°; sublimes at ≈100° when heated slowly; slight characteristic odor and slightly bitter taste; efflorescent in dry air. Anhydr cis-form: mp 104-105°; bp 258°; rapidly re-forms hydrate on exposure to air. One gram dissolves in 34 ml boiling water, 13 ml alcohol, 3 ml boiling alcohol, 135 ml chloroform, 140 ml ether, ≈1 ml boiling glacial acetic acid. At 20°, one gram dissolves in 13 ml methanol, 13 ml ethyl acetate, 250 ml water, 77 ml benzene, 290 ml carbon tetrachloride, 250 ml carbon disulfide. Practically insol in petr ether.Derivative
trans-Form.Nomenclature
CAS number: 565-50-4
Properties
Monoclinic prisms, mp 158-159°. One gram dissolves at 20° in 11 ml methanol, 20 ml ethyl acetate, 100 ml water, 250 ml benzene, 250 ml carbon tetrachloride, 500 ml carbon disulfide.Therapeutic Category
cis-Form hydrate as expectorant.
Therapeutic Category (Veterinary)
Expectorant.Keywords
Expectorant