9245. Tetrin
Description and references
Polyene antifungal antibiotic produced by Streptomyces Illinois #155-2: Pote, Diss. Abstr. 19, 2778 (1959); Gottlieb, Pote, Phytopathology 50, 817 (1960). Isoln of the two tetraenes, tetrin A and B: Rinehart et al., Ann. 668, 77 (1963); German, Diss. Abstr. 25, 97 (1964). Structure of tetrin A: Pandey et al., J. Am. Chem. Soc. 93, 3738 (1971); of tetrin B: Rinehart et al., ibid. 3747. Revised structure: R. C. Pandey, K. L. Rinehart, J. Antibiot. 29, 1035 (1976). Mode of action of tetrin A: van Etten, Gottlieb, J. Gen. Microbiol. 46, 377 (1967).
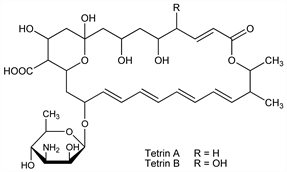
Derivative
Tetrin A.C34H51NO13; mol wt 681.77.
C 59.90%, H 7.54%, N 2.05%, O 30.51%.
Properties
Fine, colorless needles from methanol or aqueous n-butanol, mp >350° (dec). [α]D21 +8.3° (c = 0.72 in pyridine). [α]D28 +27.5° (c = 1.0 in pyridine). uv max: 214, 278, 290, 303, 318 nm (E1cm0.1% 19.4, 44.2, 81.2, 115.0, 110.9). Monobasic, pKa′ 8.30 in 60% ethanol. Sol in pyridine, dil alkalies, dil mineral acids; moderately sol in lower alcohols; practically insol in acetone, ether, water.Derivative
Tetrin B.C34H51NO14; mol wt 697.77.
C 58.52%, H 7.37%, N 2.01%, O 32.10%.