9589. Triacetin
Nomenclature
CAS number: 102-76-1
1,2,3-Propanetriol 1,2,3-triacetate; glyceryl triacetate; 1,2,3-triacetoxypropane; triacetyl glycerine; Enzactin (Ayerst); Fungacetin (Harvey). C9H14O6; mol wt 218.20.
C 49.54%, H 6.47%, O 43.99%.
Description and references
Prepd by acetylation of glycerol: Dunbar, Bolstad, J. Org. Chem. 21, 1041 (1956); by reaction of oxygen with a liquid phase mixture of allyl acetate and acetic acid using a bromide as catalyst: Keith, US 2911437 (1959 to Sinclair Refining). As an antifungal: GB 845029 (1960 to Wisc. Alumni Res. Found.). Acute toxicity: A. Wretlind, Acta Physiol. Scand. 40, 338 (1957).
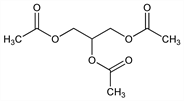
Properties
Colorless, somewhat oily liquid having a slight, fatty odor and a bitter taste. d425 1.1562, d420 1.1596, d2020 1.163. mp 78°. bp 258-260°, bp40 172°. nD20 1.4307. Sol in 14 parts water. Miscible with alcohol, ether, chloroform. Slightly sol in carbon disulfide. LD50 i.v. in mice: 1600 ±81 mg/kg (Wretlind).Use
As fixative in perfumery; solvent in manuf celluloid, photographic films. Technical triacetin (a mixture of mono-, di-, and small quantities of triacetin) as a solvent for basic dyes, particularly indulines, and tannin in dyeing.Therapeutic Category
Antifungal (topical).
Keywords
Antifungal (Synthetic)